Key Takeaways
- Cannabis law and regulations:
- Law No. 33, Decree No. 8 and Ordinance 83 regulate the medical cannabis market in Portugal.
- Licenses and authorizations available:
- Cultivation of cannabis plants.
- Wholesale trade of cannabis-based medicines, preparations and substances.
- Import and export of cannabis-based medicines, preparations and substances.
- Manufacture of cannabis-based medicines, preparations and substances.
- Marketing Authorization which allows to introduce a cannabis-based medicinal product into the market.
- Authorization for Placement on the Market which allows to place a cannabis-based preparation or substance on the market.
- Taxes and fees: Commercialization fee of 0.4% of sales volume of each medicine for which a marketing authorization was obtained.
- Market:
- As of October 2025, there were 38 companies authorized for cannabis cultivation, and several businesses registered to perform activities with controlled substances (which includes cannabis) – 26 for manufacturing, 162 for import, 158 for export, and 258 for wholesale distribution.
- According to the Portuguese Medical Cannabis Observatory, there are 12 medical cannabis substances and preparations approved for placement on the Portuguese market.
- Data provided by INFARMED shows that Portugal exported 4,850 kg of medicinal cannabis in 2020, 5,694 kg in 2021, 9,271 kg in 2022 and 11,973 kg in 2023. In 2024, cannabis exports surged by 172% to 32,558 kilograms, solidifying the country’s status as Europe’s largest exporter of cannabis.
- In 2023, 1,157 prescriptions were issued for preparations and substances based on the cannabis plant with a marketing authorization – 24.5% up from 2022.
- The International Narcotics Control Board (INCB), in its report on expected requirements of controlled substances for medical and scientific purposes by country, estimated the volume of medical cannabis required by Portugal at around 32 tonnes in 2024.
Cannabis Legalization in Portugal
Portugal decriminalized cannabis possession in 2001.
| Population (m) | 10.6 |
| GDP (€b) | 287.08 |
| Total Healthcare Expenditure (€b) | 26.56 |
| Pharmaceutical spending (% of health spending) | 14.5 |
| Alcohol Consumption (Litres Per Capita Per Year) | 10.37 |
| Prevalence of Tobacco Users (%) | 20.9 |
| Number of Cannabis Users (% of population) | 2.97 |
On July 18, 2018, Law No. 33 legalized the use of medicines, preparations and substances based on the cannabis plant for medicinal purposes and allowed for these medicines to be dispensed in pharmacies, upon presentation of a prescription.
On 15 June, 2018, the Portuguese parliament approved Decree No. 8 and the decree was then published in the official gazette on 15 January, 2019, establishing regulations to govern the cultivation, extraction, commercial preparation, local distribution, import and export, transit and sale of cannabis. An individual wishing to participate in Portugal’s medical cannabis market and perform the activities earlier mentioned, must obtain a relevant authorization from INFARMED (The National Authority of Medicines and Health Products, I.P.).
INFARMED have provided a list of conditions qualifying for medical cannabis, given that all conventional treatment methods have been unsuccessful:
- Spasticity associated with multiple sclerosis or spinal cord injuries.
- Nausea and vomiting resulting from chemotherapy, radiotherapy and combined HIV therapy and hepatitis C medication.
- Appetite stimulation in palliative care of the patient undergoing oncological or AIDS treatment.
- Chronic pain associated with oncological or nervous system conditions such as neuropathic pain caused by nerve damage, phantom limb pain, trigeminal neuralgia, or after herpes zoster infection.
- Gilles de la Tourette syndrome.
- Epilepsy and treatment of severe seizure disorders in childhood such as the Dravet and Lennox-Gastaut syndromes.
- Therapeutically resistant glaucoma.
In 2021, Ordinance 83 was published which outlined the requirements and procedures for obtaining authorizations for carrying out activities relating to the cultivation, extraction, commercial preparation, local distribution, import and export, transit and sale of cannabis.
In June 2021, during a Parliamentary debate two political parties in Portugal each presented proposals (the Left Bloc party’s Bill 859 and the Liberal Initiative party’s Bill 862) for legalization of cultivation, production, distribution, and consumption of adult-use cannabis.
On 20 April, 2023, the Liberal Initiative party filed Bill 735, proposing the legalization of adult-use cannabis. The bill would have allowed individuals over 18 years old to purchase cannabis and possess up to 25 grams. It would have also legalized self-cultivation for personal use using certain ‘authorized’ seeds.
As of December 2024 however, adult-use cannabis still has not been legalized.
Available Medical Cannabis Licenses and Authorizations in Portugal
There are 4 authorizations available for activities relating to medical cannabis: cultivation of cannabis plants, wholesale trade, import and export, and manufacture of cannabis-based medicines, preparations and substances.
Fees:
- Cultivation: $150,000
- Manufacturing: $150,000
- Wholesale: $150,000
- Import or export: $200,000
Licensing process:
The applicant must submit a request for authorization electronically to INFARMED through the Licensing Portal with the documents required for the relevant activity. The list of required documents for each activity can be found in Ordinance 83. INFARMED provides the forms and templates for submitting the application.
Information required to be submitted with the request includes:
- An application identifying the activities that the applicant wishes to perform, along with the name or corporate name and domicile or headquarters of the individual or business entity.
- If the applicant is a business entity, an updated certificate of the company’s commercial registration.
- Criminal records of the applicant obtained for the purpose of “Legal Market for Narcotic Drugs/Psychotropic Substances”.
- Short description of the project.
- Proof of implementation of security measures adopted or to be adopted, as outlined in Article 7 of Ordinance 83.
- Lease agreement for the premises of the proposed medical cannabis establishment.
- Payment of relevant fee.
In addition, there are two authorizations required for introducing or placing a cannabis-based product on the market: a Market Authorization, for medicinal products, and an Authorization for Placement on the Market, for preparations and substances. Both authorizations are issued by INFARMED.
| Marketing Authorization | This authorization is required for introducing cannabis-based medicinal products into the market. This is regulated by Decree-Law No. 176/2006. The process for obtaining the authorization and information required to be submitted are outlined on INFARMED’s website. |
| Authorization for Placement on the Market | This authorization allows to place cannabis-based preparations and substances on the market. Request fee: €1,800Request for renewal: €1,000 |
Obtaining an Authorization for Placement on the Market:
- The applicant must submit a request to the president of the highest body of INFARMED, IP, with all the required information.
- Within 90 days, INFARMED, IP, will approve or reject the applicant. The decision will be notified to the applicant and published on the INFARMED, IP, website.
Once the authorization is obtained, the holder should inform INFARMED of the price that will be charged for the authorized cannabis-based preparation or substance through the SiATS Portal in the “Cannabis for medicinal purpose” module.
It is valid for 5 years. Then, a renewal application must be submitted 9 months in advance of the authorization’s expiration date. After this first renewal, the authorization is considered to be valid for an indefinite period.
The commercialization of cannabis-based preparations or substances is also subject to a commercialization fee which is calculated as 0.4% of the sales volume of each medicine using a sales price to the reference public – this is explained in Decree-Law No. 282/95.
Cannabis Market in Portugal: Stats and Perspectives
In 2023, there was a total of 94 authorized medical cannabis businesses in Portugal; up by 38% from 2022. This number rose to 120 in 2024: a 27% increase from 2023. As of October 2025, there were 38 companies authorized for cannabis cultivation, and several businesses registered to perform activities with controlled substances (which includes cannabis) – 26 for manufacturing, 162 for import, 158 for export, and 258 for wholesale distribution.
Portugal is currently the second biggest producer of medical cannabis in the EU, after Spain, according to data from the International Narcotics Control Board (INCB). The INCB, in its report on expected requirements of controlled substances for medical and scientific purposes by country (based on data submitted by the government of each country), estimates the volume of medical cannabis required by Portugal at around 32 tonnes in 2024 which is around five times the 6.5 tonnes reported in 2023. Though this is lower than the 36 tonnes reported by Spain for 2024, professionals in the industry estimate that Portugal is actually producing twice as much medical cannabis as Spain. One agronomist has stated that Portugal has the best environmental conditions for growing cannabis in Europe.
Data provided by INFARMED shows that Portugal exported 4,850 kg of medicinal cannabis in 2020, 5,694 kg in 2021, 9,271 kg in 2022 and 11,973 kg in 2023. Almost 38% of its 2023 exports were to Germany. After Germany, the second main destination of the country’s medical cannabis exports is Spain with around 19% of total medical cannabis exported between 2020 and 2023 sent to Spain. In 2024, cannabis exports surged by 172% to 32,558 kilograms, solidifying the country’s status as Europe’s largest exporter of cannabis. The main destinations for the exported cannabis were Germany, Spain, Poland, the United Kingdom and Australia.
Portugal grows medical cannabis mainly for export and there are few cannabis-based medicinal products authorized for placement on Portugal’s market. In 2021, the first Authorization for Placement on the Market was given to Tilray’s dried flower product with 18% THC and less than 1% CBD. In the year of its authorization, Tilray’s product sold 378 packages. In 2023, the number of packages sold more than doubled to 895 boxes. Three more preparations were approved in 2024. According to the Portuguese Medical Cannabis Observatory, there are 12 medical cannabis substances and preparations approved for placement on the Portuguese market.
According to INFARMED, in 2021, 460 prescriptions for preparations and substances based on the cannabis plant with a marketing authorization were issued in Portugal. This more than doubled to 929 prescriptions in 2022 and then further rose to reach 1,157 units in 2023. From January to September 2024, 757 prescriptions were issued.
Between January and November 2024, public hospital consumption of medical cannabis reached 950 units – 34% up from the 707 units in 2023 and 81% up from the 524 units in 2022. Meanwhile, pharmacies received 1,549 packages of medicinal cannabis-based products from January to November 2024. However, due to high costs of products, many patients are still unable to receive medical cannabis treatment.
Portugal Cannabis Market Infographics
Cannabis Extraction Business Plan Sample, Portugal
'70% ready to go' business plan templates
Our cannabis financial models and cannabis business plan templates will help you estimate how much it costs to start and operate your own cannabis business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates.
Best Selling Templates
-

Cannabis Cultivation Business Plan Template
Price range: €75 through €350 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Dispensary Investor Pitch Deck Template
€75 Select options This product has multiple variants. The options may be chosen on the product page -

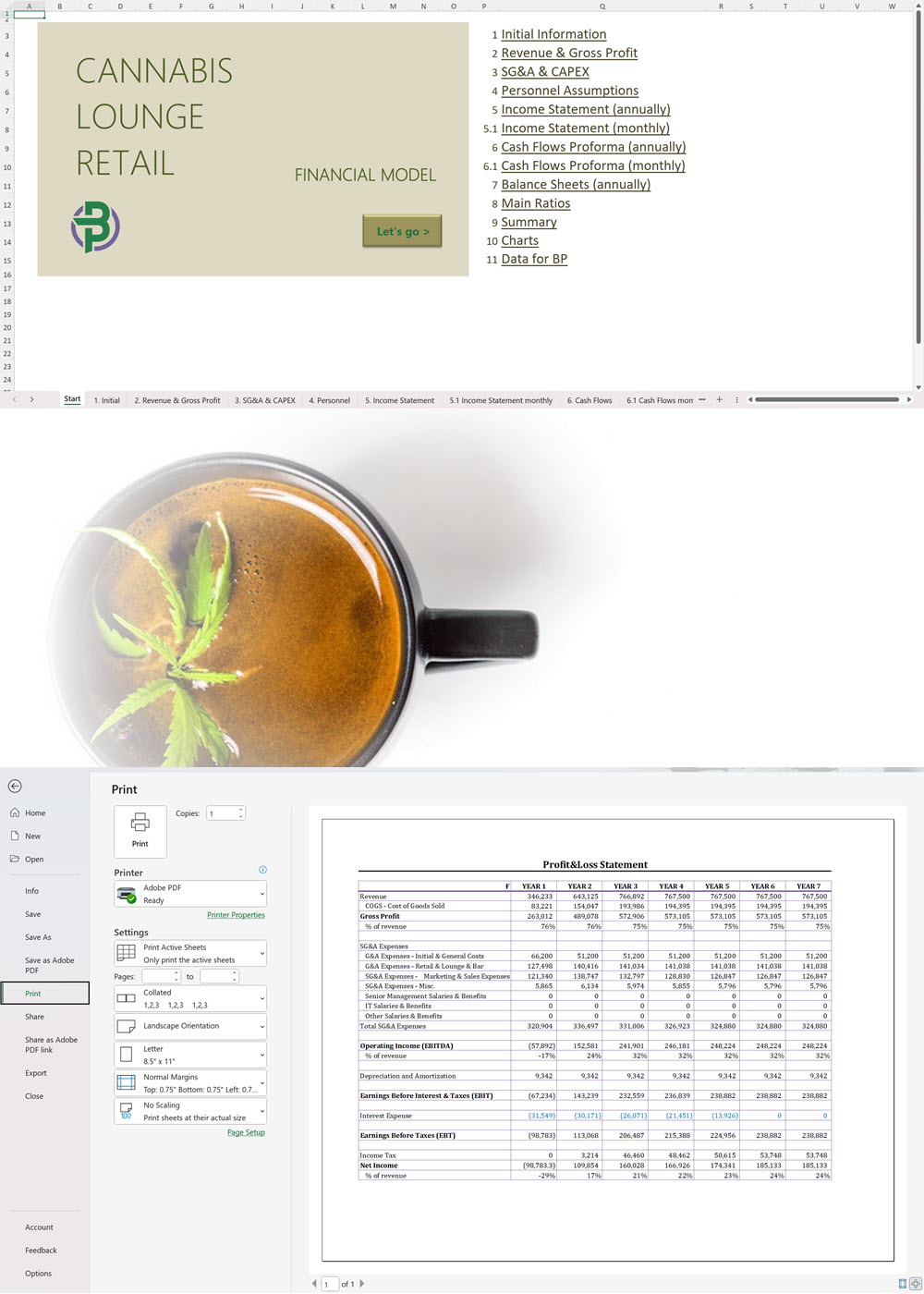
Cannabis Financial Model All in One
€250 Select options This product has multiple variants. The options may be chosen on the product page
Hemp/CBD business plan templates are also available at hempcbdbusinessplans.com.
Templates for other countries are available at cannabusinessplans.com.