1. Understand the regulatory and legal requirements. Do market research.
Cannabis Legalization
In Switzerland, medical cannabis is regulated by the Federal Act on Narcotics and Psychotropic Substances (NarcA). An amendment to this act was passed in 2011 by popular vote, allowing for the use of cannabis for medical purposes under special authorization from the Federal Office of Public Health (FOPH) on a case-by-case basis. Medical cannabis authorizations lasted for six months up until 2017, for a year from 2017 and for up to two years from 2021.
In June 2022, Switzerland’s Federal Council lifted the ban on cannabis for medical purposes by amending the Swiss Narcotics Act. As a result, from August 1, 2022, patients can get medical cannabis through a medical prescription without any exceptional permit from the FOPH. Cannabis products that are allowed to be acquired include cannabis extracts, cannabis resin, cannabis oil and cannabis tinctures. Though Switzerland has a relatively small population of patients at just over 3,000, the overall value of sales in the country is comparable to the larger markets on the continent.
In 2012, Switzerland decriminalized possession of up to 10 grams of cannabis for personal use.
On September 25, 2020, the Swiss Parliament adopted an amendment to the Federal Law on Narcotics and Psychotropic Substances. Per the amendments to Article 8a, the revised law provides a legal basis for scientific pilot trials with cannabis. Then, on 15 May 2021, the amendment came into effect allowing pilot testing of the controlled dispensing of cannabis for recreational purposes. The amendment will remain in effect for ten years and it provides the legal basis for the implementation of local and time-limited scientific pilot trials with cannabis. Applications to conduct such trials can be submitted to the Federal Office of Public Health. The rollout of legalized adult-use cannabis started in September 2022.
Multiple trials are carried out across different cities. Each trial is meant for a limited number of citizens who already consume cannabis. Trials are conducted by individual municipalities which can each supply up to 5,000 consumers at most. The purpose of the trials is to study the effects of legal access to cannabis for users in terms of health and socio-economic metrics. Volunteers can purchase personal amounts for a month’s supply. Cannabis must be both domestically and organically produced. Local manufacturers must have a production permit from the Federal Office of Public Health, ensuring quality standards.
Cannabis products that can be included in pilot trials include unprocessed products such as cannabis flowers, processed products such as cannabis extracts, cannabis products mixed with additives such as solutions containing cannabis and products intended to be smoked, vaporized, inhaled, eaten or drunk.
Pilot trials can be carried out between 2021 and 2031. The experimental protocol allows for the creation of social cannabis clubs run by universities, local authorities, research institutes, associations or foundations.
2. Decide the type of cannabis business. Choose a location. Check local zoning regulations.
Cannabis Licenses Available in Switzerland
There are two kinds of cannabis license categories available in Switzerland:
- Medical cannabis licenses issued by Swissmedic.
- Recreational cannabis licenses issued by the FOPH to cultivate, manufacture or import cannabis for use in Swiss pilot trials.
Those responsible for a pilot trial select their growers, producers and manufacturers themselves so applicants wishing to cultivate cannabis must not only apply for a license from the FOPH, but also contact the organization responsible for a pilot trial for which they want to cultivate, manufacture or import cannabis or cannabis products. The FOPH acts as a coordinator of the authorization procedure. The interested applicant must submit an application for an exceptional license and the organization conducting the pilot trial for which the cannabis cultivated, manufactured or imported by the applicant will be used must submit a corresponding application to the FOPH.
It is also possible for the applicant to communicate their interest to the FOPH so that they are recorded on a list of interested producers and FOPH will then give the list to those responsible for a pilot trial.
Activity | Licenses required |
| Medical cannabis | |
| Manufacturing, distribution, import, export and trade of medical cannabis | An establishment license for handling controlled substances allows to manufacture, procure, distribute, import, export and trade medical cannabis. A pharmacy does not require to hold an establishment license for handling controlled substances unless it makes more than 9 distributions of medicines containing controlled substances in a calendar year. An establishment license is valid for up to 5 years. A renewal application must be submitted at least six months before the expiration date. Also, an import or export permit is required when importing or exporting medical cannabis. A one-time import or export permit is valid for up to 4 months. A general import or export permit is valid until the end of the current calendar year at maximum. Each permit is given for a single import or export and for a specific maximum quantity. It is only valid within the framework of the business relationship between the supplier and the recipient. |
| Cultivation of medical cannabis | Two licenses are required.
|
| Recreational cannabis | |
| Cultivation | An exceptional cultivation license allows to cultivate cannabis for use in Swiss recreational cannabis pilot trials. |
| Manufacturing | An exceptional manufacturing license allows to manufacture cannabis products for use in Swiss recreational cannabis pilot trials. |
| Import | An exceptional import license allows to import cannabis and cannabis products for use in Swiss recreational cannabis pilot trials.An import license from Swissmedic is also required. Cannabis and cannabis products can only be imported in exceptional circumstances if sufficient quantity cannot be produced in Switzerland on a timely basis. |
3. Develop a solid cannabis business plan. Secure financing.
4. Prepare required documents and obtain the required licenses.
Licensing Process:
A detailed guide on the license and permit requirements for each activity can be found on Swissmedic’s website.
To obtain an establishment license, the applicant must:
- Be entered in the commercial register.
- Ensure correct storage in accordance with Art. 54 NarcCo.
- Have a Responsible Person for controlled substances who must be a healthcare professional or have a science degree.
- Submit the following information:
- First and last name of the applicant is they are an individual. Designation according to the commercial register if the applicant is a business establishment.
- First and last name, date of birth and function of the person responsible for the controlled substances.
- Applicant’s place of residence or business address.
- List of controlled substances the applicant wishes to handle.
- Type of activity the applicant wishes to perform.
- Cultivation and storage location (including customs warehouse and bonded warehouse).
- Current and complete commercial register extract.
- Private extract from the criminal record information system VOSTRA of the responsible person, not older than six months.
- Professional qualifications and the professional career of the responsible person.
- Submit an application for an establishment license for the cultivation of cannabis for medical purposes to Swissmedic.
- Swissmedic will review the submitted application documents and the Canton will carry out an inspection.
- Once an establishment license for the cultivation of cannabis for medical purposes has been obtained, the applicant can apply for an individual cultivation license through the NDS-Web Portal.
To obtain an establishment license, the applicant must:
- Be entered in the commercial register.
- Ensure correct storage in accordance with Art. 54 NarcCo.
- Have a Responsible Person for controlled substances who must be a healthcare professional, have a science degree or diploma issued by a university or university of applied sciences in agriculture, environmental sciences or forestry.
- Ensure adequate protection against theft and prevention of use for other purposes.
Documents required to be submitted to Swissmedic when applying for an individual cultivation license include:
- First and last name or designation of the applicant.
- A traceability and quality assurance system for medical cannabis.
- A written supply agreement with details on the type and quantity of cultivation and the obligation of the purchasing party to accept the delivery of the harvest.
- Type of cultivation that will take place in the specific cultivation cycle such as whether it will be indoor/outdoor, what seeds will be used, variety that will be cultivated with its expected THC content, exact location and size of the cultivation area and the expected yield in kilograms.
Information required to be submitted with the application:
- First and last name of the cultivator or the company name.
- First and last name as well as vocational qualifications and career history of the person responsible for cultivation and their deputy.
- Extract from the criminal excerpt for the responsible person and their deputy, issued within six months.
- Which pilot trial cannabis will be cultivated for and the type and quantity of cannabis that the applicant intends to grow.
- Number of seeds and seedlings and the purchase location.
- Precise types of plants requested, including the expected yield, THC and CBD content and total THC (in grams).
- Desired duration of the license.
- Description of cultivation areas.
- Description of storage, protection and logging of seeds/plants/crops policies.
Information required to be submitted with the application:
- Company name as written in the commercial register.
- First and last name as well as vocational qualifications and career history of the person responsible for manufacture and their deputy.
- Which pilot trial cannabis products will be manufactured for and the type and quantity of cannabis products that the applicant intends to manufacture.
- Desired duration of the license.
- Annual production volume.
- Supplier of cannabis raw material.
- Required quantity of cannabis raw material, type, THC and CBD content and total THC (in grams).
- Description of storage, protection and logging of cannabis products policies.
Information required to be submitted with the application:
- Description of the pilot trial and exact import address.
- First name and last name of the person responsible for the import and their deputy.
- Certificate of need for the import and proof that sufficient quantities cannot be produced in Switzerland in a timely manner.
- Supplier, including exact address.
- Information on the type of cannabis or cannabis products imported.
- Import quantities.
- Desired duration of the license.
5. Keep track of your ongoing compliance requirements.
Cannabis Advertising in Switzerland
Advertising of controlled substances to the public is prohibited. If medical cannabis or medical cannabis products are advertised to medical professionals, they must be clearly designated as a medicinal product subject to control under the Narcotics Act.
How to Start a Cannabis Business in Switzerland? Infographics.
How Can Cannabusinessplans Templates Help Start or Grow Your Cannabis Business?
The majority of cannabis licensing programs require the submission of a business plan as part of the application for a cannabis license. Even if it is not mandated by law, a business plan is essential for securing funding and assisting an applicant in understanding how much money it will take to start a cannabis business and how much profit it could make.
A well-structured business plan can make all the difference in helping you demonstrate your proposed cannabis business’ compliance with regulations and its operational viability.
Our complete cannabis business plan template package is everything you need to create a professional business plan for a cannabis business with expert financials and projections. A complete cannabis business plan template package includes:
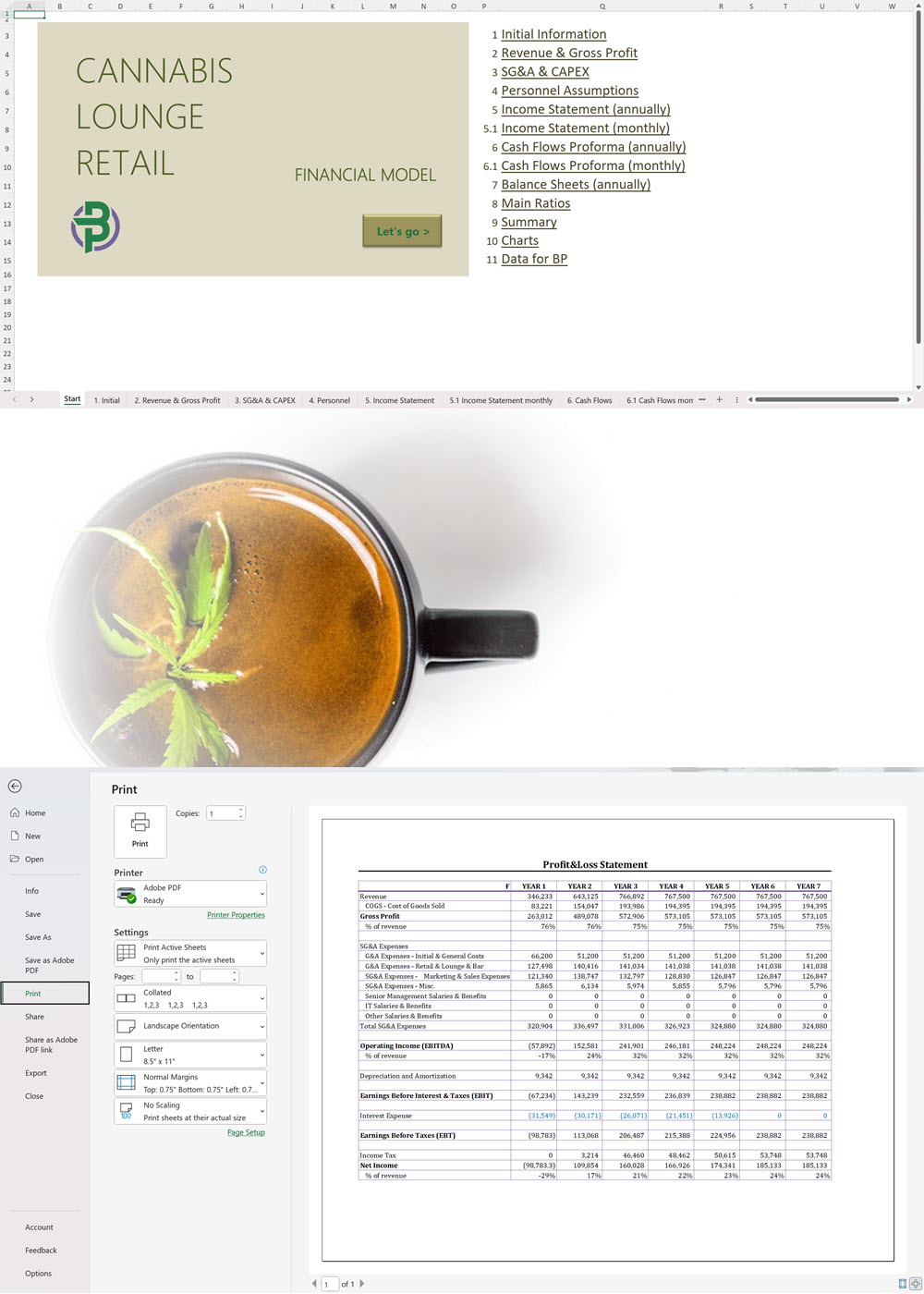
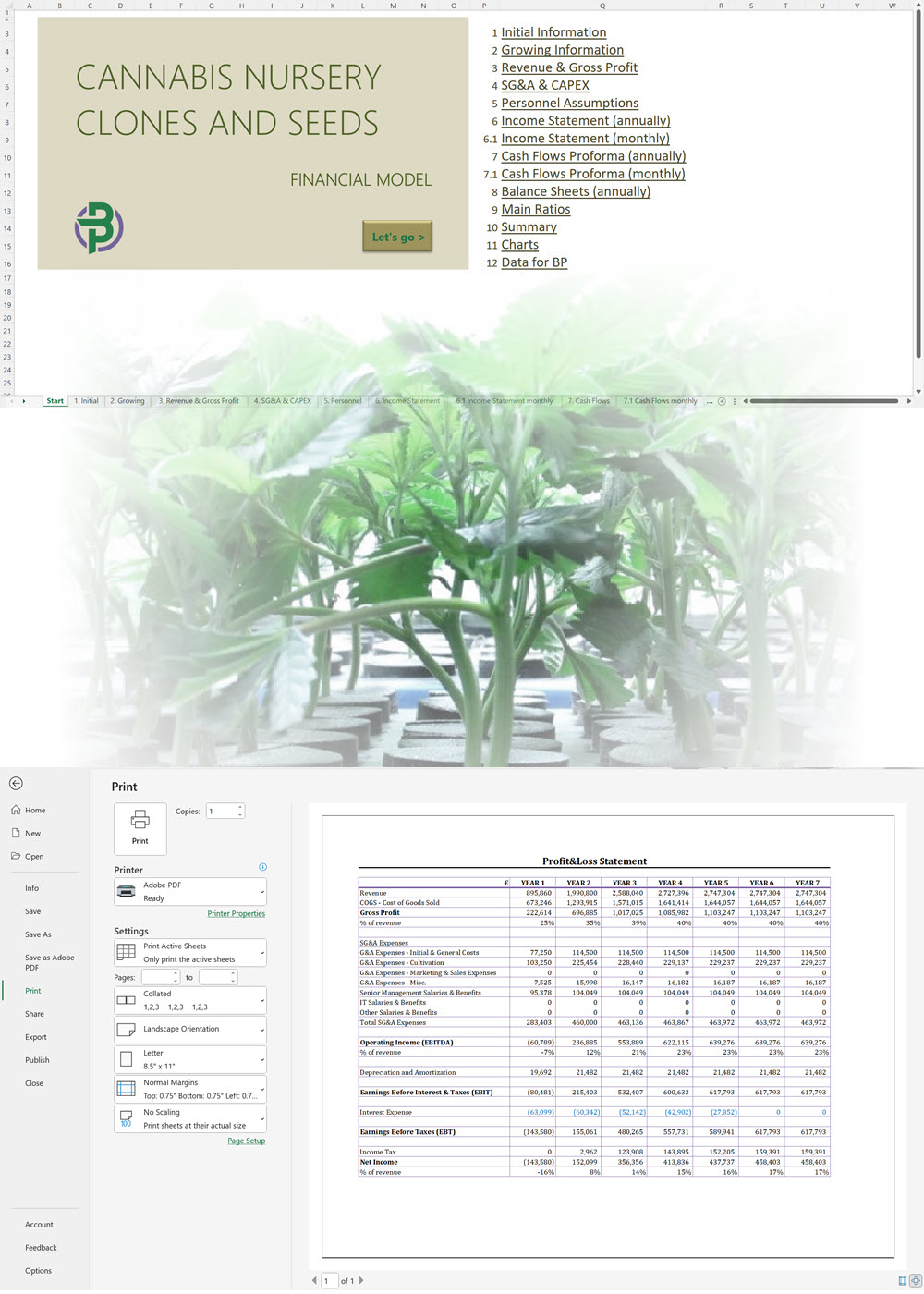
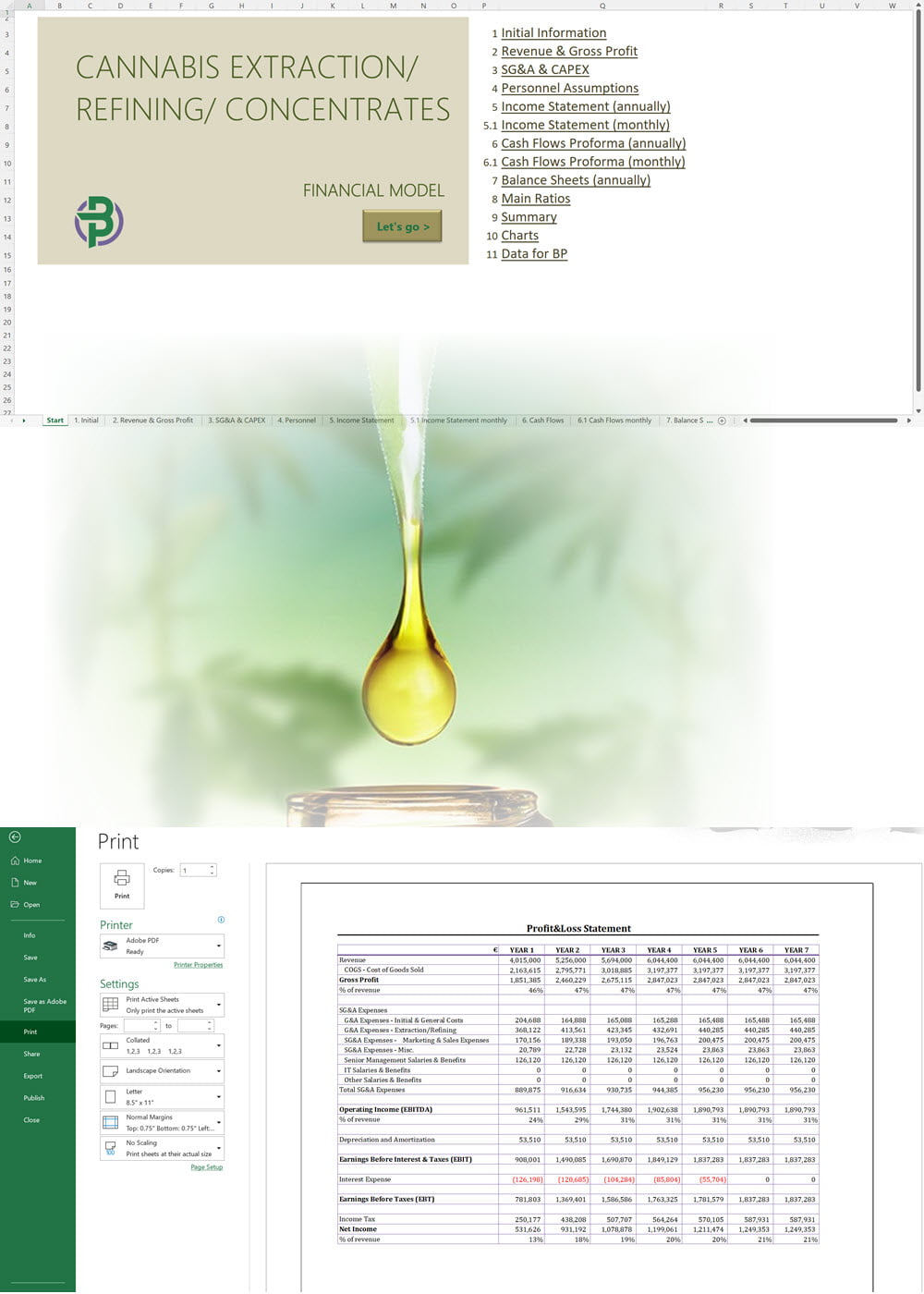
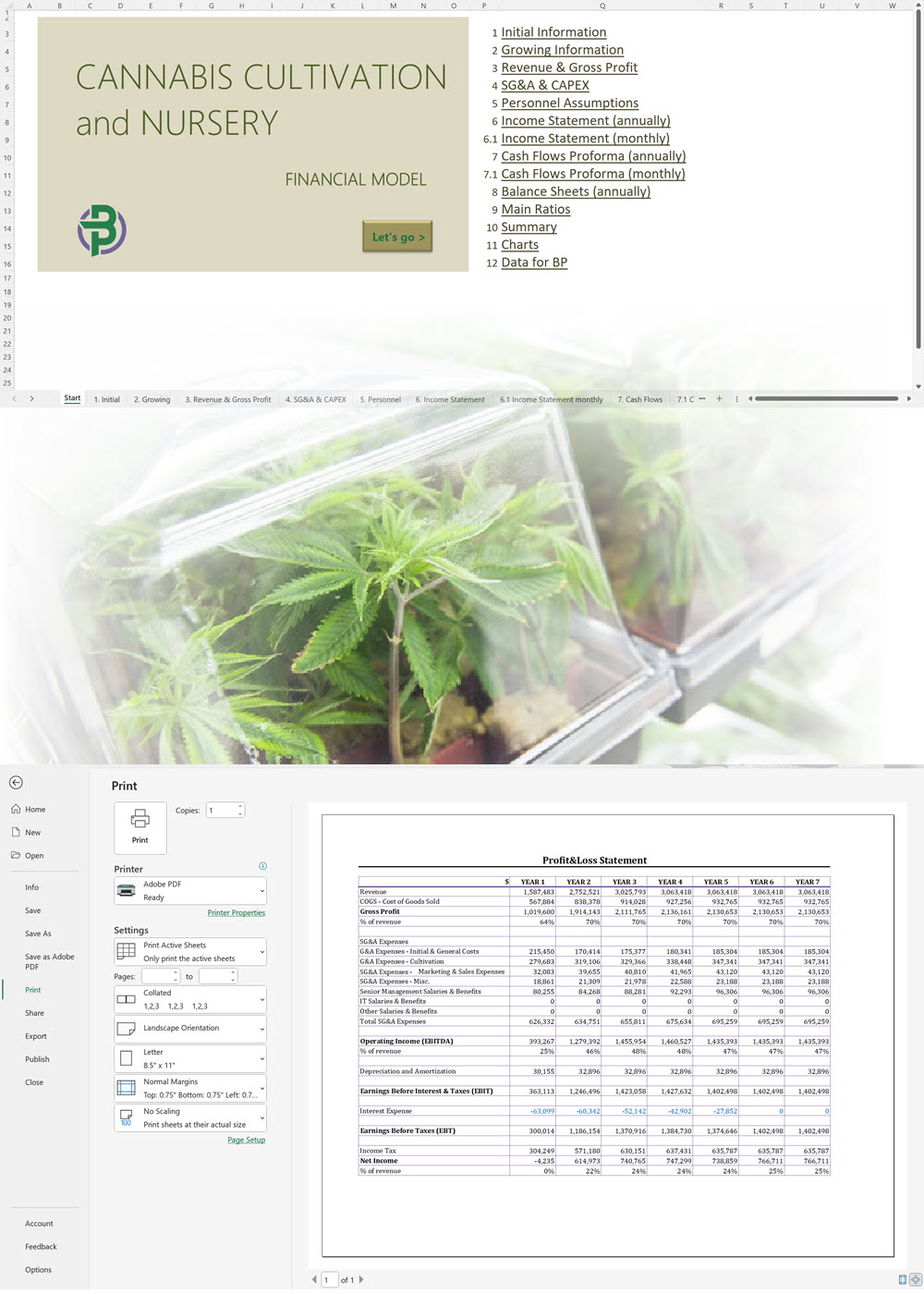
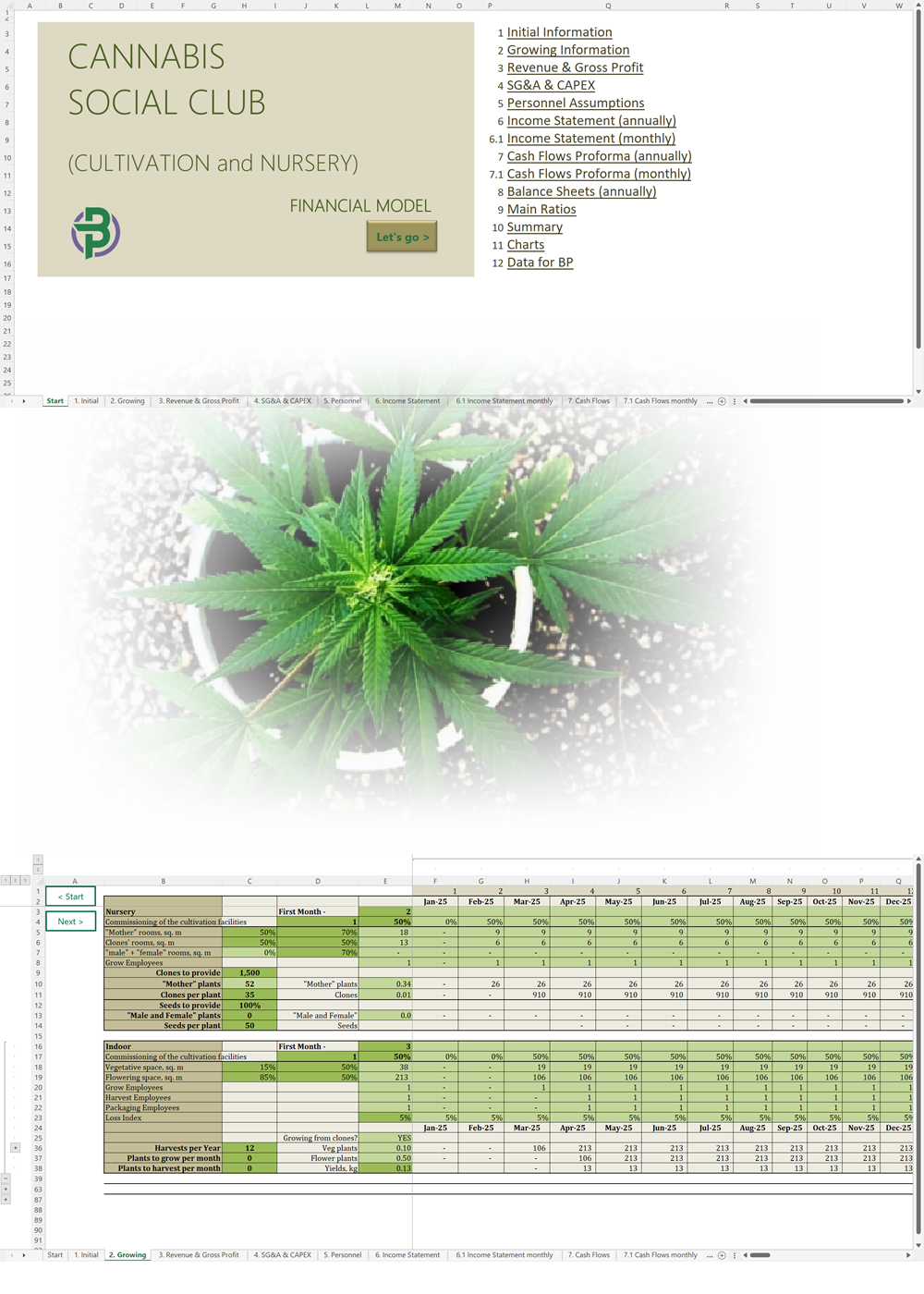
- Excel Financial Model: change variables and immediately see the impact, break down operational and capital costs, know how much it will take to get into the business and the potential profits.
- Word Business Plan: value proposition, market analysis, marketing strategy, operating plan, organizational structure, financial plan and more.
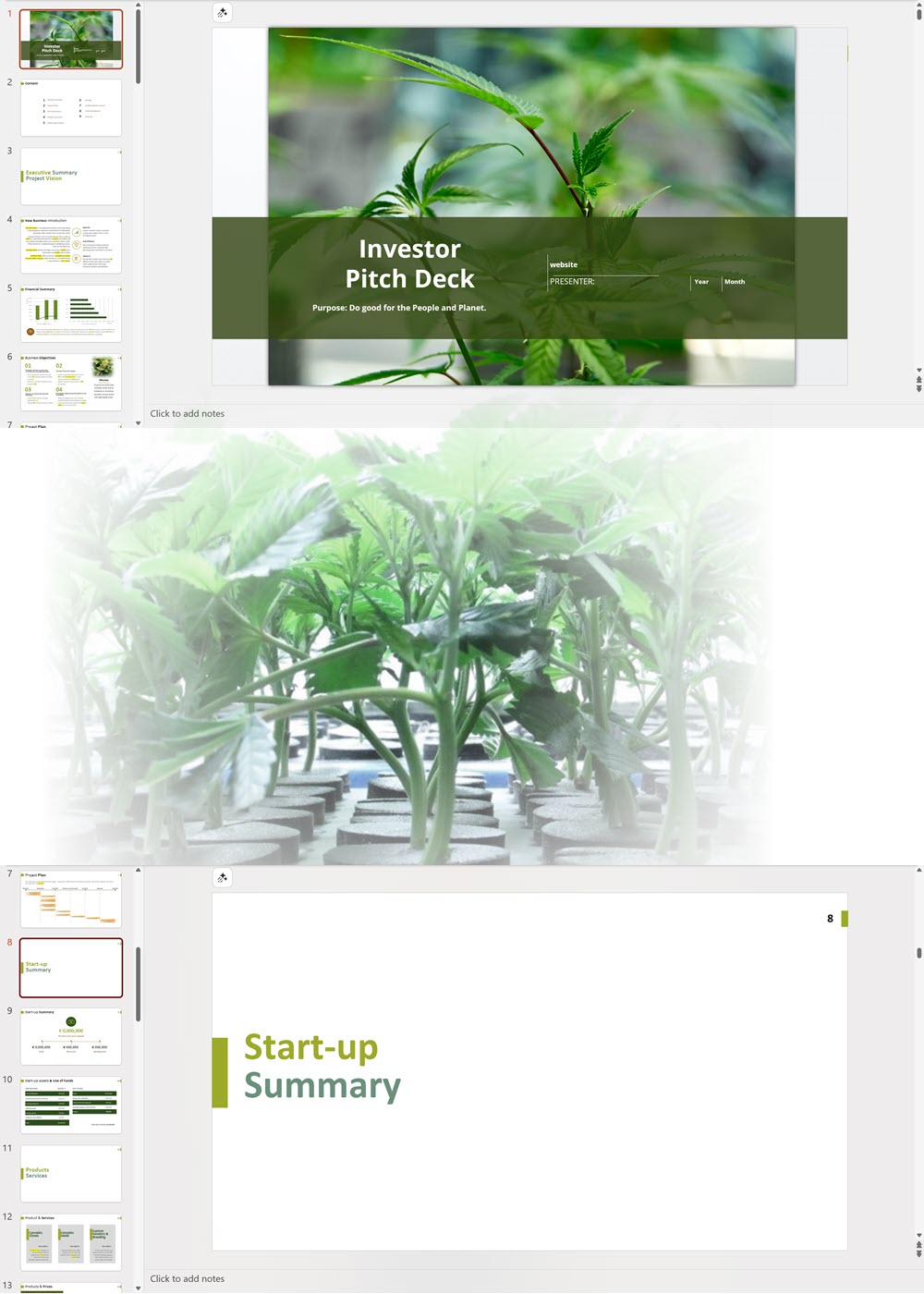
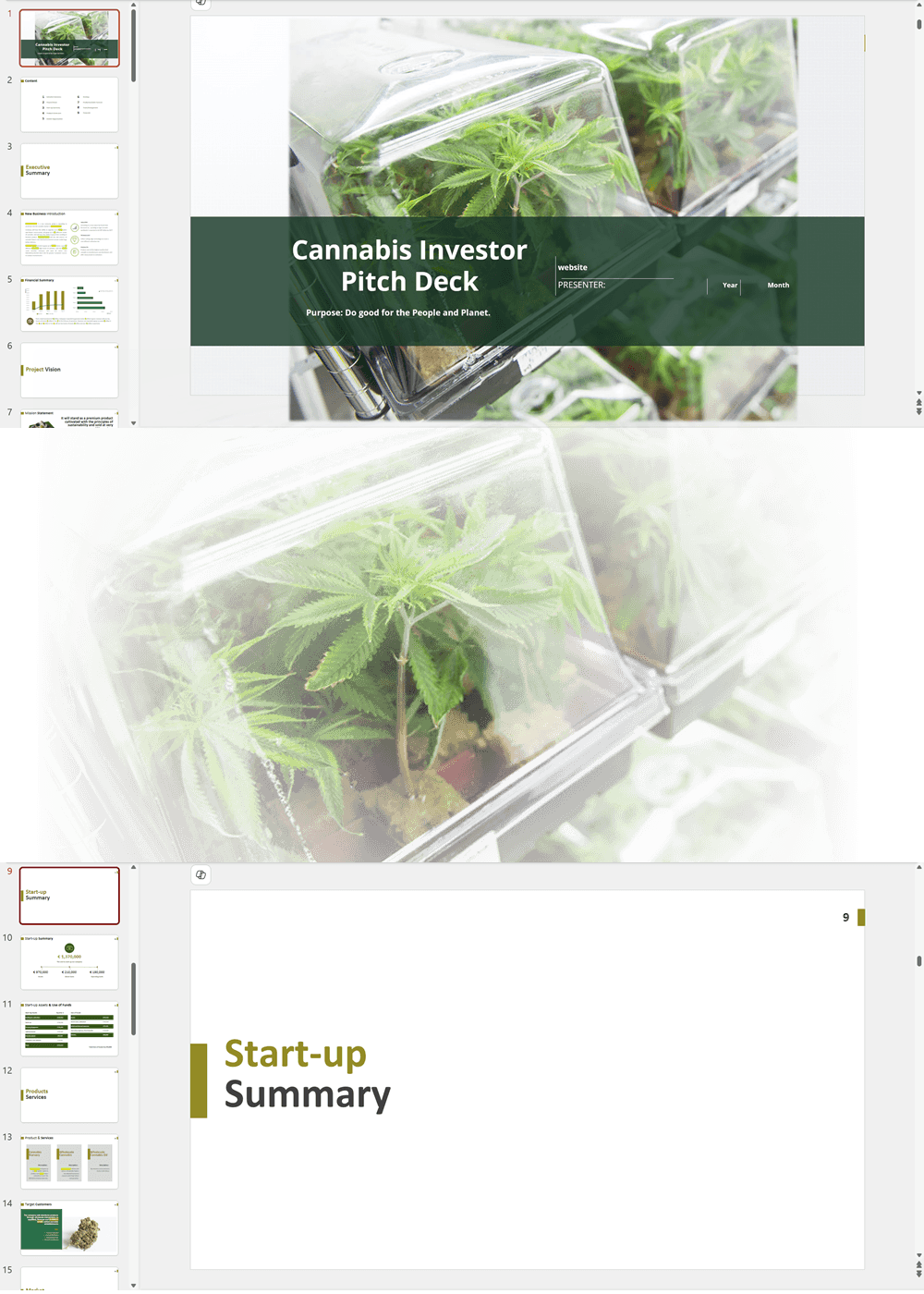
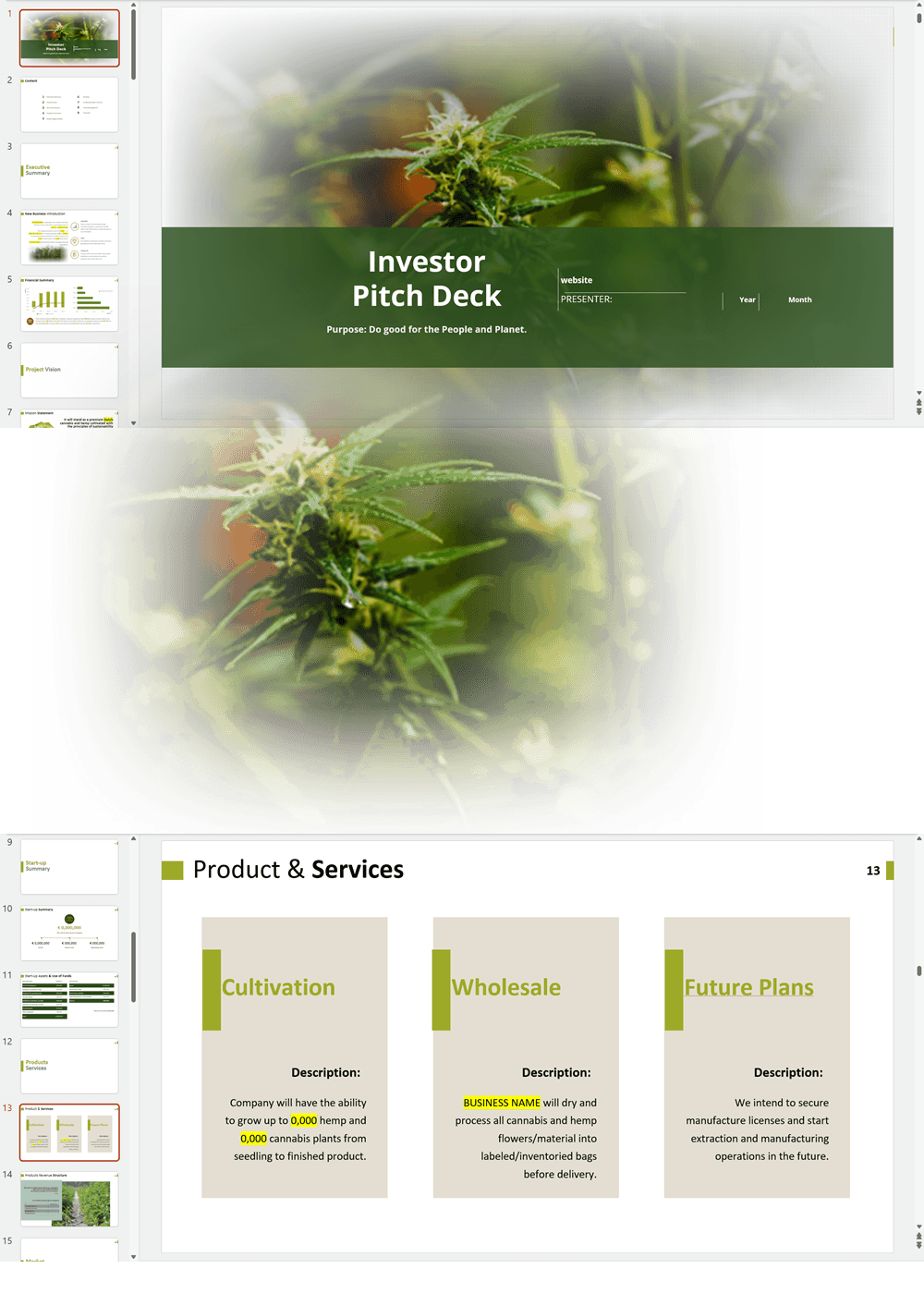
- PowerPoint Pitch Deck: provide a quick overview of your cannabis business plan.
'70% ready to go' business plan templates
Our cannabis financial models and cannabis business plan templates will help you estimate how much it costs to start and operate your own cannabis business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates.
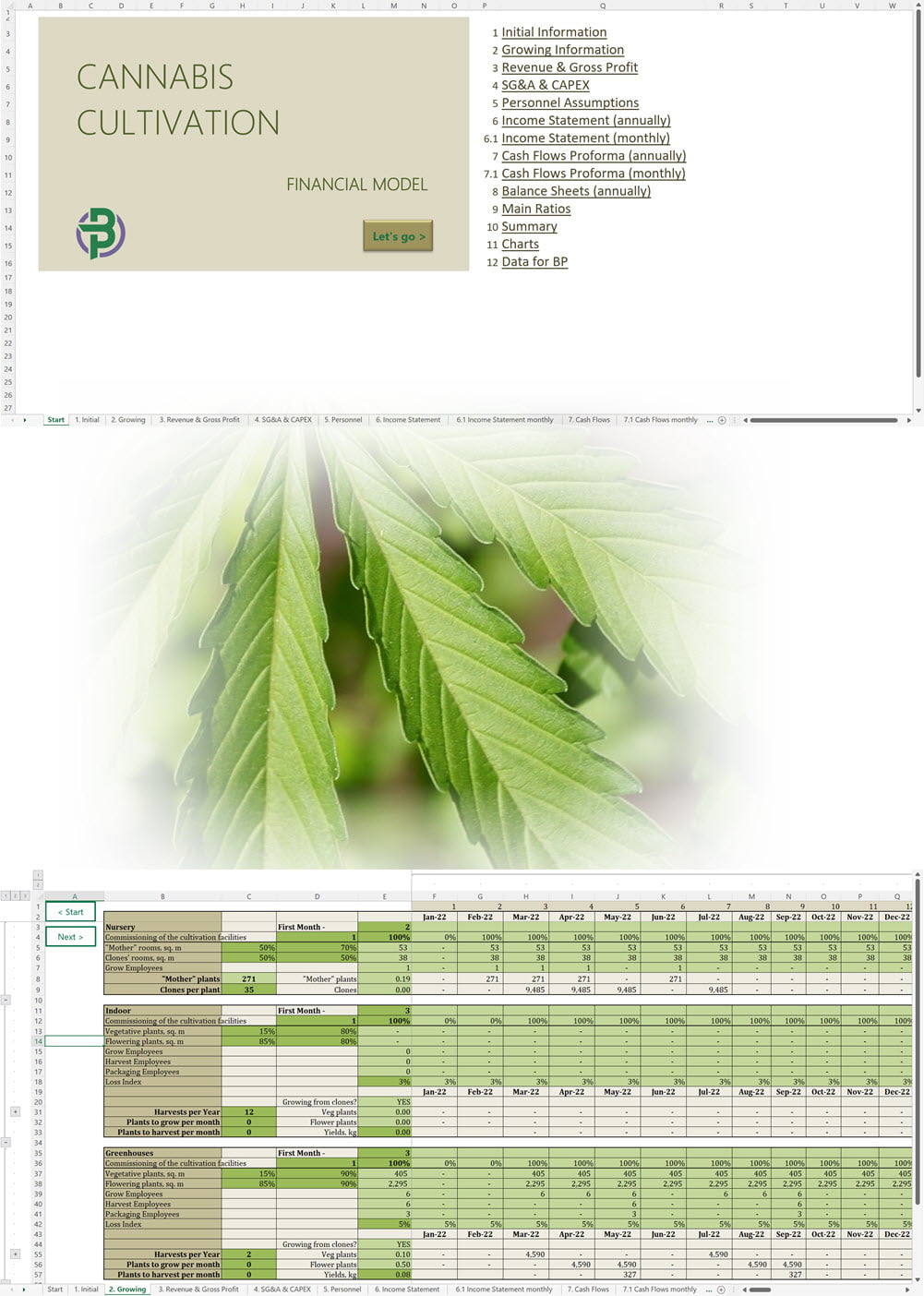
Cannabis Cultivation Business Plan Sample, Switzerland
Best Selling Templates
-

Cannabis Cultivation Business Plan Template
Price range: €75 through €350 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Dispensary Investor Pitch Deck Template
€75 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Financial Model All in One
€250 Select options This product has multiple variants. The options may be chosen on the product page
Hemp/CBD business plan templates are also available at hempcbdbusinessplans.com.
Templates for other countries are available at cannabusinessplans.com.