Cannabis Laws in France
In 2013, France legalized the sale of medications containing cannabis derivatives under prescription and only when no other treatments were effective in helping the patient. Prior to this, cannabis-related activities were prohibited in France.
| 2025 Population (m) | 68.63 |
| 2025 GDP (€b) | 2,738 |
| 2024 Total Healthcare Expenditure (€b) | 333 |
| 2023 Total Beauty & Well being Expenditure (€b) | 15 |
| Cumulative Number of Patient Enrollments in Medical Cannabis Pilot Programme | 3,209 |
| Healthcare Practitioners Trained under Medical Cannabis Pilot Programme | 2,291 |
New regulations were introduced by the National Assembly on 23 November 2018 to reduce penalties for possession of personal amounts. In the same year, a multidisciplinary scientific committee was formed to research medical cannabis and in December 2018, this committee identified the need for medical cannabis to be available to patients in certain clinical situations.
Medical Cannabis Pilot Scheme
On October 25, 2019, France’s National Assembly gave approval for a medical cannabis pilot scheme, to be supervised by the National Agency for the Safety of Medicines and Health Products (ANSM). The project was at first planned to last for two years: from March 26, 2021, until March 26, 2023. However, the pilot program was extended by one year by Article 57 of Law No. 2022-1616 on December 23, 2022. After the end of the program, the authorities planned to review the results and consider a widening of accessibility to medical cannabis.
Under this project, up to 3,000 patients with certain qualifying conditions and who have not responded to other treatments, were able to participate in the trial and access medical cannabis free of charge. Before France passed a decree in 2022, allowing the domestic cultivation of cannabis, medical cannabis for this pilot project was imported from abroad.
In October 2020, the regulators released a statement announcing the opening of the application process to secure tenders to supply the pilot program, specifying a number of THC:CBD ratios as well as a variety of permitted product formats. Formats could be dried flower, capsules and oils. The plan was to select two suppliers for each of these product categories; one as the main supplier and the second as a substitute if there are supply disruptions.
In 2021, the following producers were selected by the French authorities – Aurora Cannabis (Canada, main supplier), Tilray (Canada, main supplier), Little Green Pharma (Australia, main supplier), Panaxia (Israel, main supplier), Althea (Australia, substitute supplier), Emmac Life Sciences (the UK, substitute supplier). The selected producers had to supply cannabis products to participating companies at their own cost.
In February 2024, the ANSM stopped prescribing medical cannabis in flower form.
A total of 3,209 people participated in France’s medical cannabis pilot program and 1,849 were still receiving treatment, as of 18 September, 2024. Nearly 1,500 pharmacies and 744 doctors signed up to participate in the experiment.
Data collected during the two years of the pilot program demonstrated an operational distribution system and the effectiveness of cannabis in helping patients, with few serious adverse effects. Therefore, the Social Security Financing Act for 2024 established that cannabis-based medicines should be made available for use by patients, provided that these medicines are first authorized by the ANSM.
From 27 March, 2024, no new patients were allowed to enter the pilot program. Initially, the French Ministry of Health extended the availability of cannabis-based medicines for patients participating in the experiment until March 31, 2026. However, in January 2026 this deadline was further extended with no exact duration set for this extension.
As the deadline kept being extended, the number of patients still participating in the program fell dramatically to just 700, indicating that many patients have possibly become tired of waiting for a formal, permanent medical cannabis framework to be implemented.
Domestic Supply Chain
On March 1, 2022, a new decree went into effect which allows the cultivation, manufacturing, import and distribution of cannabis-based medicines in France as long as cultivators and companies in the supply chain are authorized by the Director General of the ANSM. The decree also states that only cultivators who have a supply contract with a pharmaceutical establishment or other establishment authorized by the ANSM can hold and cultivate cannabis plants for use in production of cannabis-based medicines. The ANSM oversees the establishment of the medical cannabis supply chain.
Medical Cannabis Legalisation Progress
In March 2025 the French government submitted the decree implementing Article 78 of the Social Security Financing Act for 2024, the Ministerial Order on Cultivation and Production, and the Ministerial Order on Technical Specifications to the European Commission for approval. In June 2025, the decree and two orders were approved by Brussels and then in August 2025 they were validated by the Conseil d’État (state Council). They must now be signed by the Ministries of Health and Economy and published in the Journal Officiel.
If the proposed framework is approved, medical cannabis medicines would have to receive a five year authorization granted by the ANSM before they can be made available to patients. Only pharmaceutical establishments authorized under Articles L. 5124-1 and L. 5138-1 of the Public Health Code would be allowed to apply to ANSM for cannabis cultivation, import, or export authorizations. Cultivators would be required to grow cannabis only indoors and have an existing binding contract with an authorized pharmaceutical establishment before they can begin to grow cannabis.
Medical cannabis products would be available in the form of flower as well as oral and sublingual formulations, and would be split into three ratio profiles: THC-dominant, balanced and CBD-dominant. Flower will be available for vaporization only, supplied in sealed, single-use dosages.
The Haute Autorité de Santé (HAS) is responsible for designing a price and reimbursement structure, determining prescriber training requirements, and drafting other recommendations. The HAS cannot finalize their price-setting and training requirements until the decrees are officially published in the Journal Officiel.
As of April 2026, the decrees have not been signed yet by the Ministries of Health and Economy or published in the Journal Officiel. Formal adoption of the decrees is expected to occur in June 2026 while HAS reimbursement final opinion is anticipated in October-November 2026. Some industry experts now estimate that full market implementation will take place some time in 2027.
How to Obtain a Cannabis Authorization in France
If an individual wishes to cultivate, manufacture, distribute or perform other activities with cannabis in France, they must send an email to the address on the website of ANSM, requesting an activity authorization on a relevant form provided.
Cannabis cultivators must comply with Good Agricultural Practices. Manufacturers must comply with EU Good Manufacturing Practices and demonstrate, through a technical dossier, that its extraction and formulation processes allow it to produce finished products that are safe, reproducible, and meet the specifications set by the French Ministry of Health.
The technical dossier must include:
- Technical description of the extraction process.
- Validated analytical controls to accurately measure cannabinoids and detect any contaminants.
- 12 months stability data.
- Documentation relating to the traceability of each batch.
For import or export authorizations, the individual must also send an email to the ANSM using the form provided by ANSM. Each import application must include:
- The characteristics of the imported product.
- The name of the manufacturer.
- The country of origin.
- The justification for medical use.
- The recipient’s contact details.
Medical Cannabis Market in France
According to a French doctor who is the president of the scientific committee responsible for monitoring the medical cannabis pilot program, based on the five medical conditions which warranted participation in the pilot project, between 150,000 and 300,000 people in France could be prescribed medical cannabis. According to data from Prohibition Partners, among the 1,655 patients participating in the pilot program in June 2025, around 60% had neuropathic pain as their qualifying condition. The second most frequent qualifying condition was multiple sclerosis with 247 patients reporting it, followed by epilepsy (reported by 154 patients), palliative treatment (105 patients), cancer treatment (93 patients) and painful spasticity of other pathologies excluding MS (59 patients).
The French Monitoring Centre for Drugs and Drug Addiction (OFDT) found that in 2021, 47.3% of adults aged between 18 and 64 in France, reported that they have used cannabis in their lifetime. OFDT’s survey in 2022 also found that around 10.6% of the population have used cannabis in the past year.
Prohibition Partners estimates that the medical cannabis patient numbers in France could climb from 5,454 in 2026 to 457,129 in 2035, and project that a regulated medical cannabis market could reach €806 million in annual revenue by 2035.
However, the potential value of France’s medical cannabis market can actually be greater considering the fact that an established medical-only cannabis market in Florida has nearly 1 million patients as of August 2025 – 4.3% of its total population – bringing in around $2.4 billion annually in revenue. Applying this percentage to France’s population, there would be an estimated number of nearly 3 million potential medical cannabis patients in the country. From this it follows that France’s medical cannabis market, once fully developed, can be worth $7.2 billion (€6.19 billion) – three times as much as Florida’s.
How Can Cannabusinessplans Templates Help Start or Grow Your Cannabis Business?
The majority of cannabis licensing programs require the submission of a business plan as part of the application for a cannabis license. Even if it is not mandated by law, a business plan is essential for securing funding and assisting an applicant in understanding how much money it will take to start a cannabis business and how much profit it could make.
A well-structured business plan can make all the difference in helping you demonstrate your proposed cannabis business’ compliance with regulations and its operational viability.
Our complete cannabis business plan template package is everything you need to create a professional business plan for a cannabis business with expert financials and projections. A complete cannabis business plan template package includes:
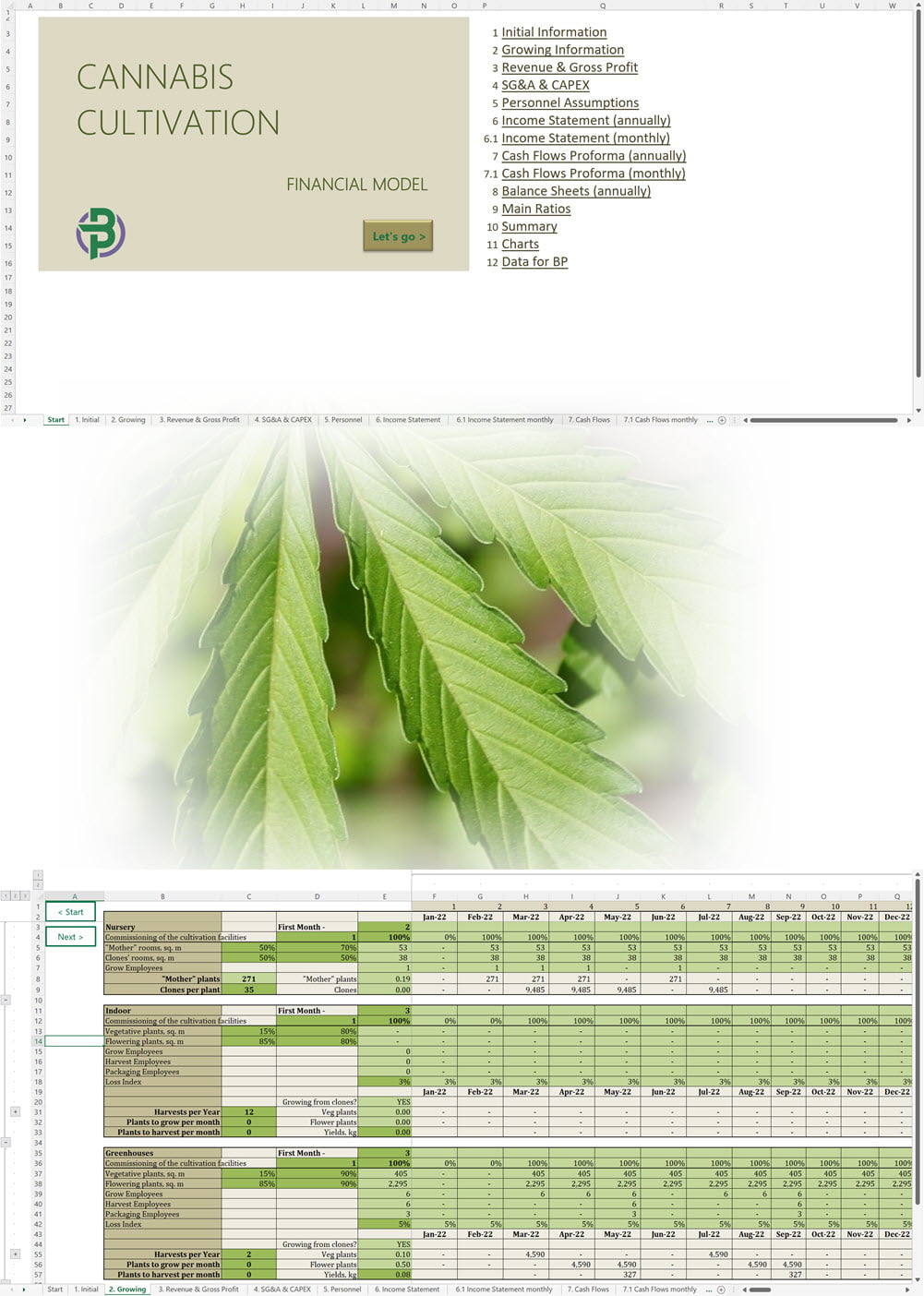
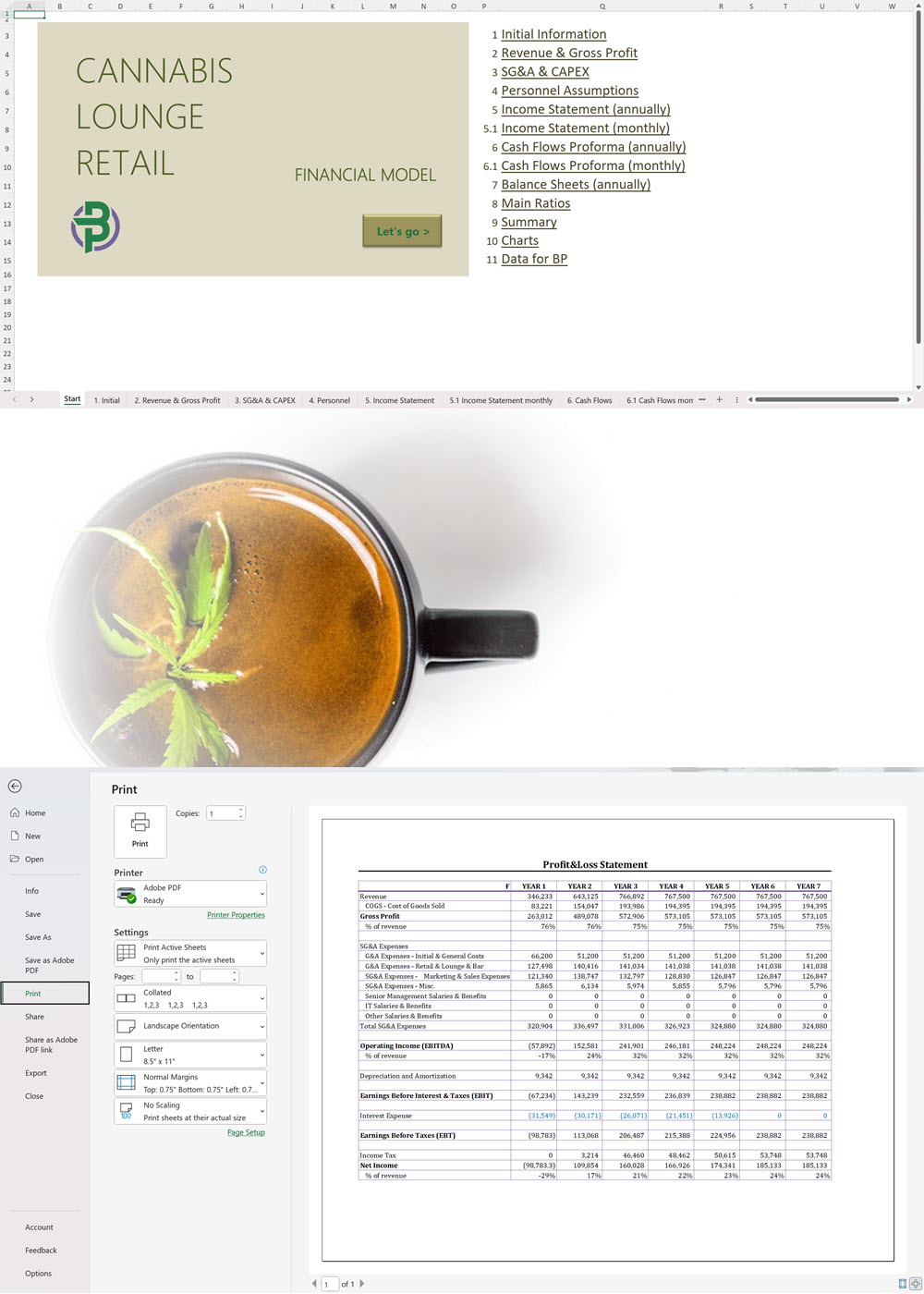
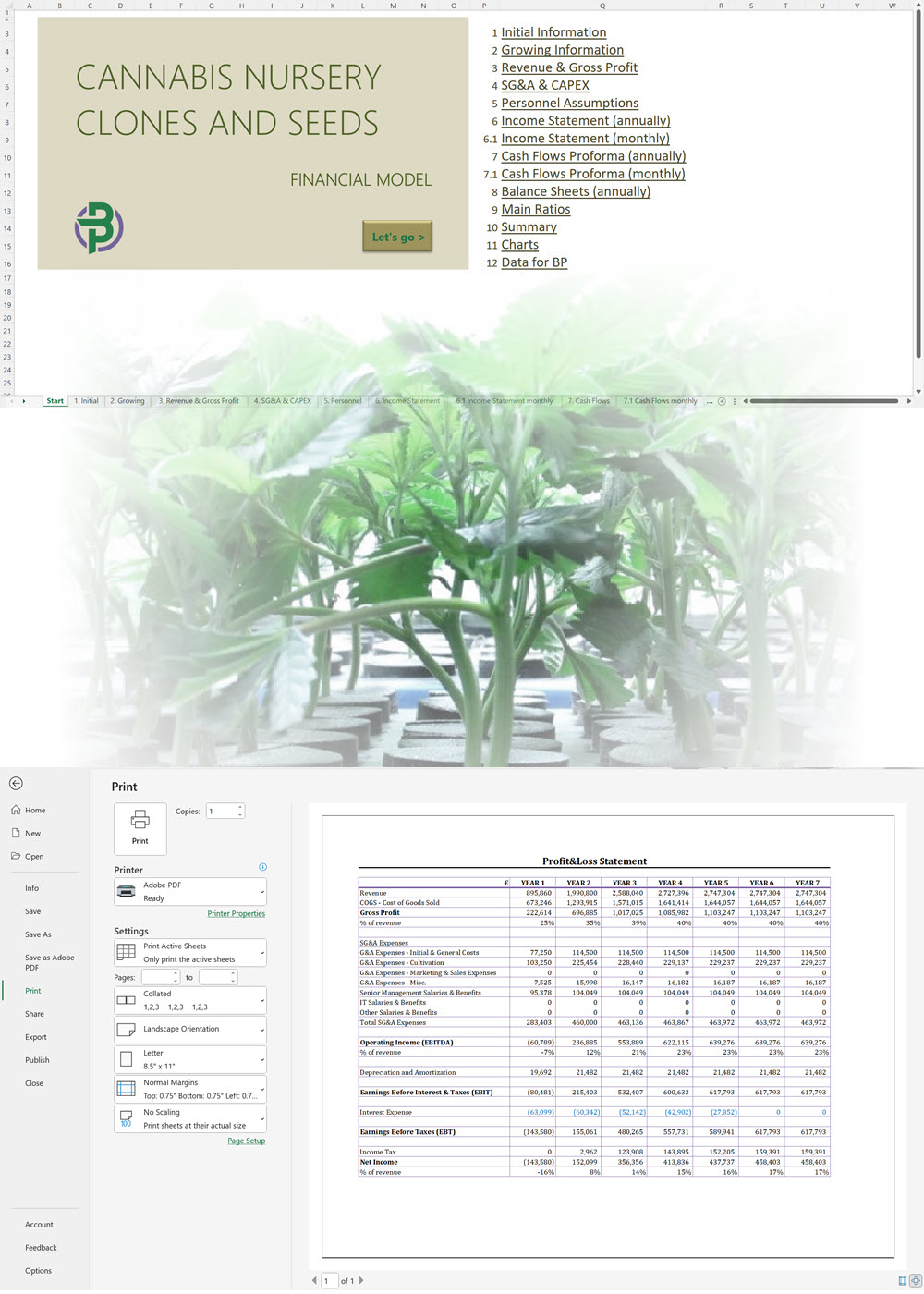
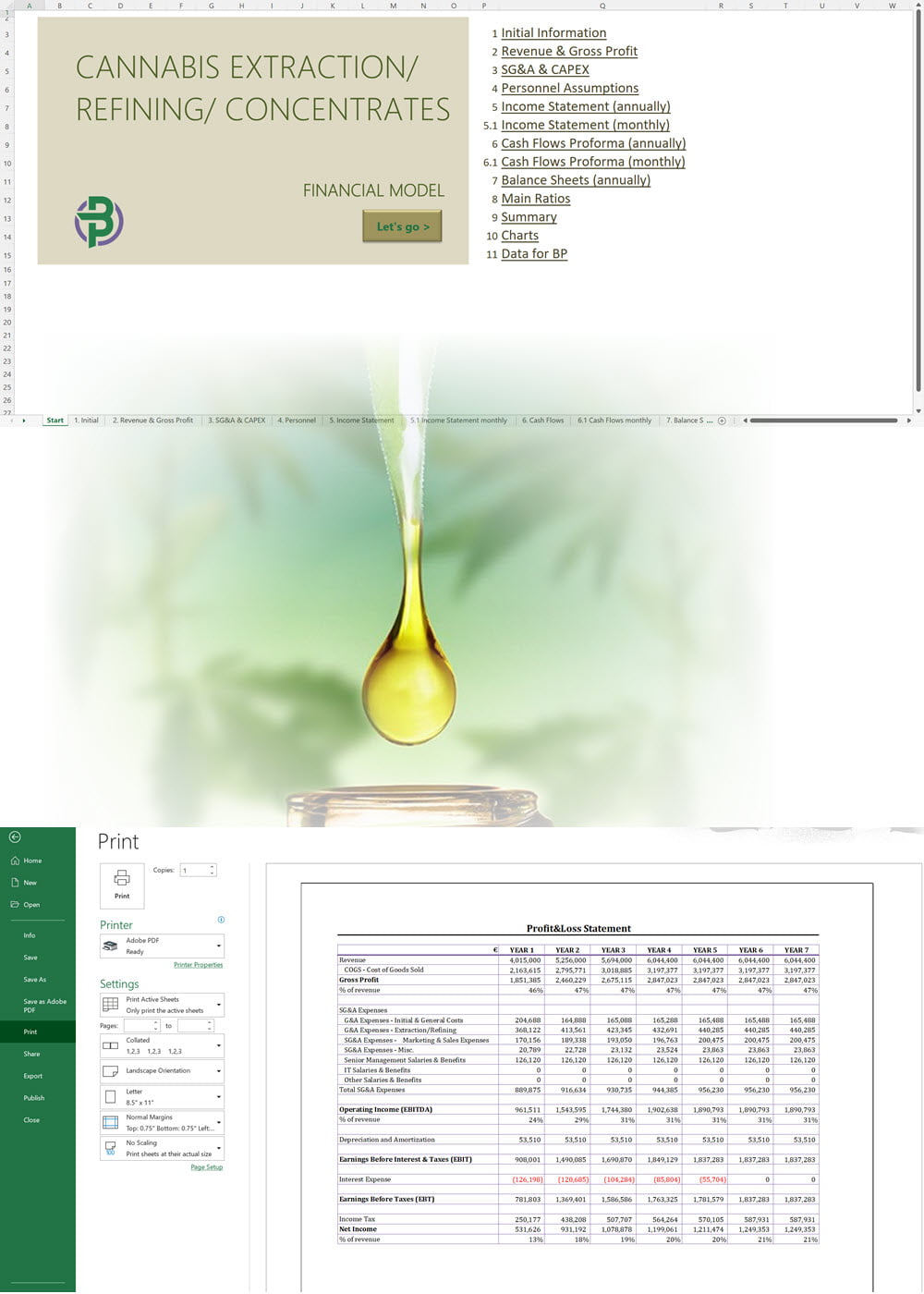
- Excel Financial Model: change variables and immediately see the impact, break down operational and capital costs, know how much it will take to get into the business and the potential profits.
- Word Business Plan: value proposition, market analysis, marketing strategy, operating plan, organizational structure, financial plan and more.
- PowerPoint Pitch Deck: provide a quick overview of your cannabis business plan.
'70% ready to go' business plan templates
Our cannabis financial models and cannabis business plan templates will help you estimate how much it costs to start and operate your own cannabis business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates.
Cannabis Manufacturing (with in-house processing) Business Plan Sample, France
Best Selling Templates
-

Cannabis Cultivation Business Plan Template
Price range: €75 through €350 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Dispensary Investor Pitch Deck Template
€75 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Financial Model All in One
€250 Select options This product has multiple variants. The options may be chosen on the product page
Cannabis business plan templates for other countries can be found at cannabusinessplans.com.