Key Takeaways:
- Hemp Industry Regulations:
- The industrial hemp industry in Italy is regulated by Law 242/2016 while the use of hemp for medical purposes is regulated by Presidential Decree 309/1990.
- Authorizations available:
- Authorization to cultivate hemp plants from certified seeds.
- Authorization to manufacture narcotic and psychotropic substances – allows to manufacture cannabis extracts with pharmacologically active substances.
- Products legal:
- Industrial hemp products which include food, cosmetics, raw materials, biodegradable and innovative semi-finished products for various industries. All products must contain no more than 0.2% THC, though up to 0.6% is tolerated. Hemp leaves and inflorescences are prohibited from use in edible products.
- Industry stats:
- Hectares of hemp cultivated in Italy grew by 900% from just 400 hectares in 2013 to 4,000 hectares in 2017.
- 904 tonnes of hemp seeds for planting were imported in 2022. The largest supplier was Ethiopia, accounting for 83% of the imported hemp seeds.
- 902 tonnes of hemp seeds for planting were exported in 2022. The three main destinations were France (45% of the exported hemp seeds), Germany (27%) and Austria (17%).
Hemp Legalization
Hemp cultivation and processing for production of food, cosmetics, raw materials, biodegradable and innovative semi-finished products for various industries became legal in Italy on December 2, 2016, through law No. 242/2016. This law permitted industrial hemp cultivation without the need for prior authorization, provided that certified seeds with THC no higher than 0.2% are used. However, natural variations resulting in no more than 0.6% THC are tolerated (farmers are not penalized if THC content is not higher than 0.6%). Hemp leaves and inflorescences are prohibited from use in edible products. Hemp growers must only plant hemp varieties registered in the EU’s “Common Catalogue of Varieties of Agricultural Plant Species”. They must also keep the labels of hemp seed purchased for at least one year. In January 2022, a Ministerial Decree tried to limit hemp production to seeds and derivatives only and to classify leaves and inflorescences as narcotics. However, on February 14, 2023, the Regional Administrative Court of Lazio ruled that the entire plant, including floral parts, can be used industrially and reaffirmed that the whole hemp plant is non-narcotic. An authorization from the Italian Ministry of Health is required in order to cultivate and manufacture products from hemp-derived CBD for pharmaceutical use.
Authorization for Hemp Cultivation for Pharmaceutical Use
Two authorizations must be obtained from the Ministry of Health’s Central Office for Narcotics in order to cultivate hemp and manufacture cannabis extracts that are to be transferred to pharmaceutical factories and used in preparation of pharmaceuticals. The first authorization is for the cultivation of hemp plants from certified seeds, pursuant to art. 27 of Presidential Decree 309/1990. It permits cultivation and supply of leaves and inflorescences to pharmaceutical companies authorized (by the relevant national authority of the country where the company is located) to manufacture active pharmaceutical ingredients and which possess a Good Manufacturing Practices (GMP) certification. The authorization is granted to agricultural companies. The applicant must have an agreement with the pharmaceutical company for exclusive transfer and supply of hemp between the two parties. The second authorization is for the manufacture of narcotic and psychotropic substances, pursuant to art. 32 of Presidential Decree 309/1990. Only pharmaceutical companies authorized to manufacture pharmacologically active substances by the Italian Medicines Agency can apply for this authorization. It permits the manufacture of cannabis extracts with pharmacologically active substances. Both authorization applications and the transfer agreement must be submitted at the same time to the Central Narcotics Office. Outlined below are procedures and requirements for each of the two authorizations:
Authorization for the cultivation of hemp plants from certified seeds
Fee: €229.70 Two revenue stamps: €16.00 each.
- Submit the application form with all the required documentation to the PEC address indicated here, and pay the fee and revenue stamps online through the Ministry of Health’s platform.
- Within 90 days after the date of the application submission, the applicant will be notified via e-mail whether their authorization has been granted or denied.
Documentation required to be submitted according to the authorization form:
- Certificate of registration with the Chamber of Commerce with the name of the owner or legal representative, the company name and the location of the company.
- Description of the species and variety of cultivation allowed by the European Union legislation.
- Detailed description of the cultivation activity for which the authorization is requested and an estimate of the quantity of starting plant material planned to be produced annually.
- Operating procedures for personnel management.
- Location, cadastral parcels and surface area of land (in hectares) on which cultivation will take place.
- Indication of the locations where starting plant material will be loaded and unloaded and the storage procedure for semi-finished products or products ready for transfer to a pharmaceutical company.
- Security and surveillance systems in place.
- Technical report with a floor plan of the product storage area as well as a description of anti-intrusion systems in place.
- Personal data and qualification of the individual responsible for Good Agricultural and Collection Practice (GACP) control of the cultivation with their registration number in the Register of Agronomists and Foresters.
- Company name, legal representative, address, PEC and telephone numbers of the authorized pharmaceutical company to which hemp will be supplied.
- Agreement for the supply of hemp to the authorized pharmaceutical company.
- Declaration of GACP cultivation according to European Union legislation signed by the person responsible for GACP cultivation, the person responsible for the agricultural company and the person responsible for the pharmaceutical company.
- Certificate relating to the criminal record of the owner or legal representative of the company as well as the person responsible for GACP cultivation.
- Certificate relating to pending charges from the Public Prosecutor’s Office at the Court of the owner or legal representative as well as the person responsible for GACP cultivation.
- Certificate relating to the residence and family status of the owner or legal representative as well as of the person responsible for the GACP cultivation.
- Acceptance of the assignment by the person responsible for the GACP cultivation.
- Certificate of the educational qualification of the person responsible for GACP cultivation.
Authorization for the manufacture of narcotic and psychotropic substances
Fee: €229.70 Two revenue stamps: €16.00 each. Procedure:
- Submit the application form with all the required documentation to the PEC address indicated here, and pay the fee and revenue stamps online through the Ministry of Health’s platform.
- Within 90 days after the date of the application submission, the applicant will be notified via e-mail whether their authorization has been granted or denied.
Documentation required to be submitted according to the authorization form:
- Copy of authorization issued by AIFA for the production of raw materials with the name of the qualified person(s).
- Certificate of registration with the Chamber of Commerce with the name of the owner or legal representative, the company name and the location of the company.
- Certificate relating to the criminal record of the owner or legal representative of the company as well as the qualified person.
- Certificate relating to pending charges from the Public Prosecutor’s Office at the Court of the owner or legal representative as well as the qualified person.
- Certificate relating to the residence and family status of the owner or legal representative as well as of the qualified person.
- Self-certification of the qualifications of the qualified person.
- Acceptance of the assignment by the qualified person.
- Premises plan indicating the place where narcotics will be stored.
- Building features of the place where the warehouse is located with descriptions of the premises and a summary of the security measures used for effective storage of narcotic or psychotropic substances and preparations, policy for staff access to the warehouse and any insurance against theft taken out by the company.
The authorization is valid for two years. Three months before the expiration date, a renewal request must be submitted on the same form as the initial application for the authorization and with all the same documents except for the floor plan of the premises and the related technical report.
Importing Hemp
According to Article 189 of the EU Regulation No. 1308/2013, an import license is required for all hemp imports. Hemp seeds for planting must be accompanied by proof that the variety contains no more than 0.3% THC. Hemp seeds not intended for planting can be imported by authorized importers only who must submit proof that the seeds are in a condition that does not allow them to be used for planting. Raw true hemp must have no more than 0.3% THC.
Hemp Market
In 2013, only around 400 hectares of hemp was cultivated in Italy but after the passage of Law 242/16, it grew by 900% to 4,000 hectares in January 2017. According to a USDA report on the Italian hemp industry, Italy imported 904 tonnes of hemp seeds for planting in 2022 – 61% up from 2021 and 262% up from 2020. The largest supplier was Ethiopia, accounting for 83% of the imported hemp seeds. Meanwhile, 902 tonnes of hemp seeds for planting were exported in 2022 – an 11% increase from 2021 and a dramatic 5,538% increase from 2020. The three main destinations were France (45% of the exported hemp seeds), Germany (27%) and Austria (17%). Regarding hemp seeds for purposes other than planting, in 2022, 828 tonnes and 346 tonnes were imported into Italy and exported out of Italy, respectively. The tonnes of raw true hemp imported and exported in 2022 were much more modest – 62 tonnes and 18 tonnes, respectively. After the passage of Law 242/2016, several shops (called ‘cannabis light’ stores) appeared, selling hemp inflorescences and its derivatives, including edibles, cosmetics and other hemp products. As of 2023, there were around 2,000 ‘light cannabis’ stores in Italy. However, hemp inflorescences and its derivatives are not among the products authorized by Law 242/2016, making these shops illegal. On July 31, 2024, the joint Constitutional Affairs and Justice voted to amend Law 242/2016 and ban all flowering cannabis, including hemp inflorescences (hemp flowers, stems and stalks). In response, several Italian hemp industry associations have sent a petition to the European Commission (EC). The EC accepted the petition and plans to carry out a ‘preliminary investigation into the issue’.
Source: hempcbdbusinessplans.com
Italian Hemp Industry Infographics
Hemp Cultivation and Processing Business Plan Sample, Italy
'70% ready to go' business plan templates
Our hemp/CBD financial models and business plan templates will help you estimate how much it costs to start and operate your own hemp/CBD business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates.
Best Selling Hemp Templates
-

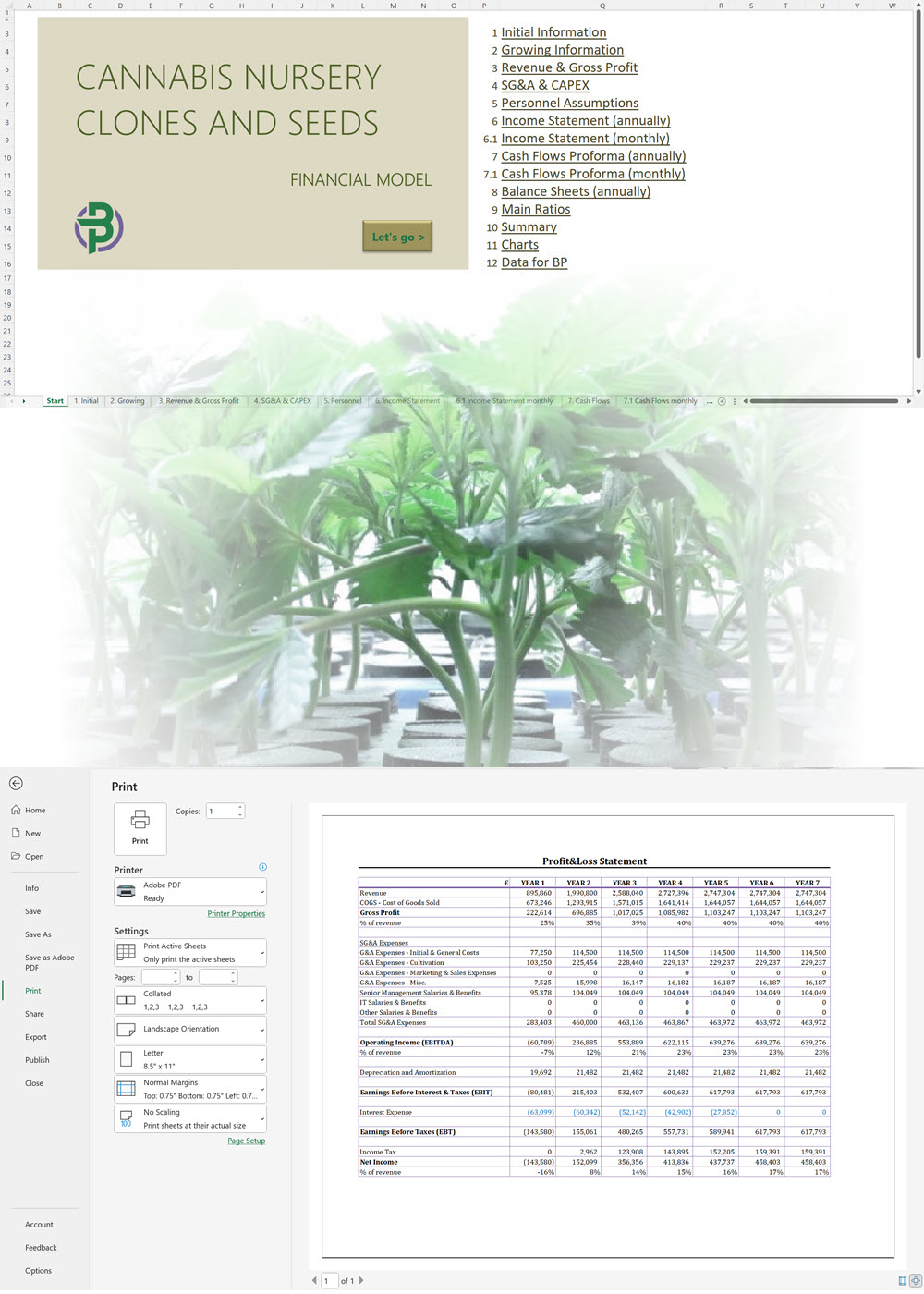
Hemp Cultivation + CBD Oil Extraction and/or Fiber Products Financial Model
€150 Select options This product has multiple variants. The options may be chosen on the product page -

CBD Retail Investor Pitch Deck Template
€70 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis and Hemp Vertically Integrated Business Plan Template
Price range: €100 through €550 Select options This product has multiple variants. The options may be chosen on the product page
For more information on the hemp industry and hemp/CBD business plan templates, visit hempcbdbusinessplans.com.