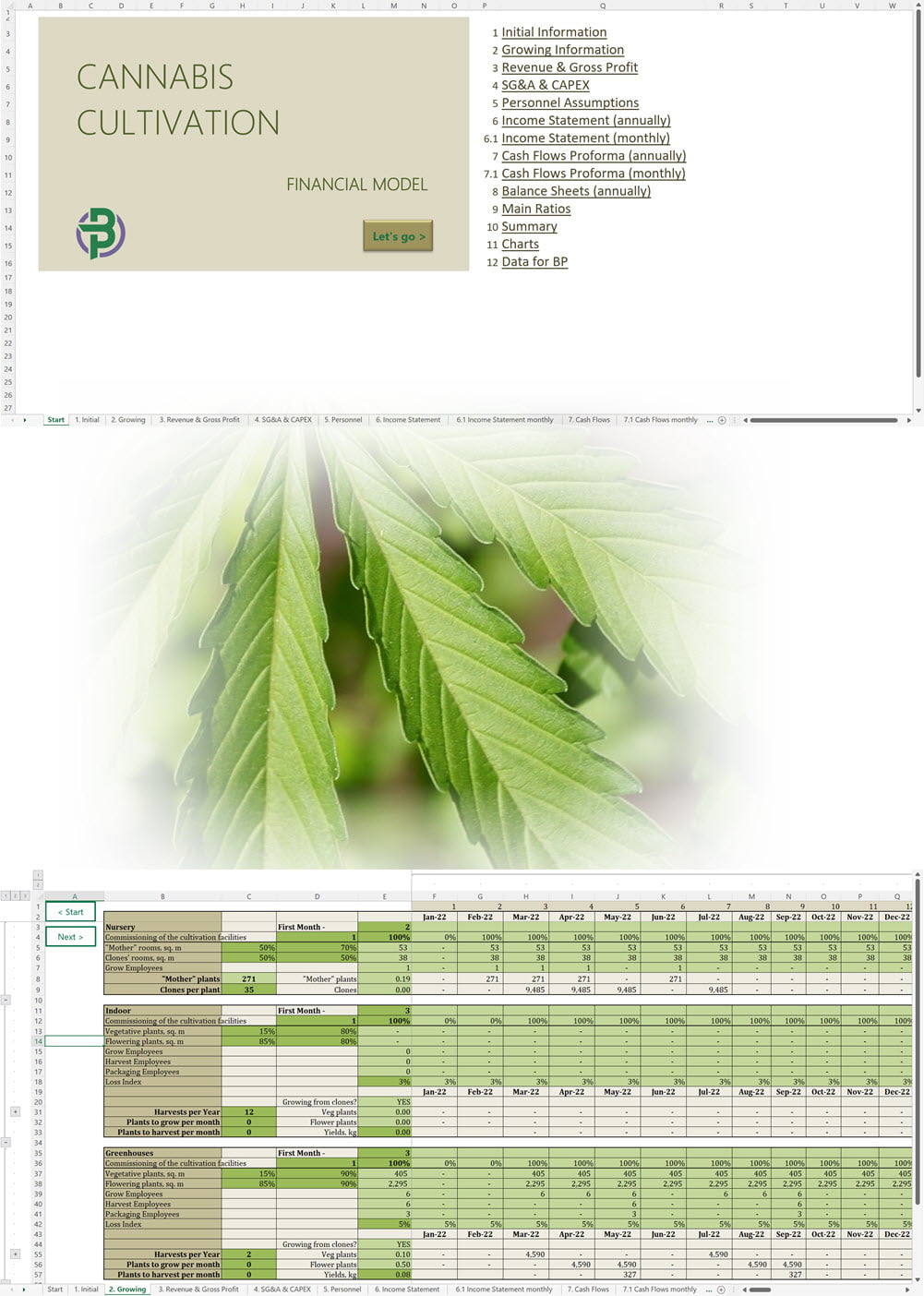
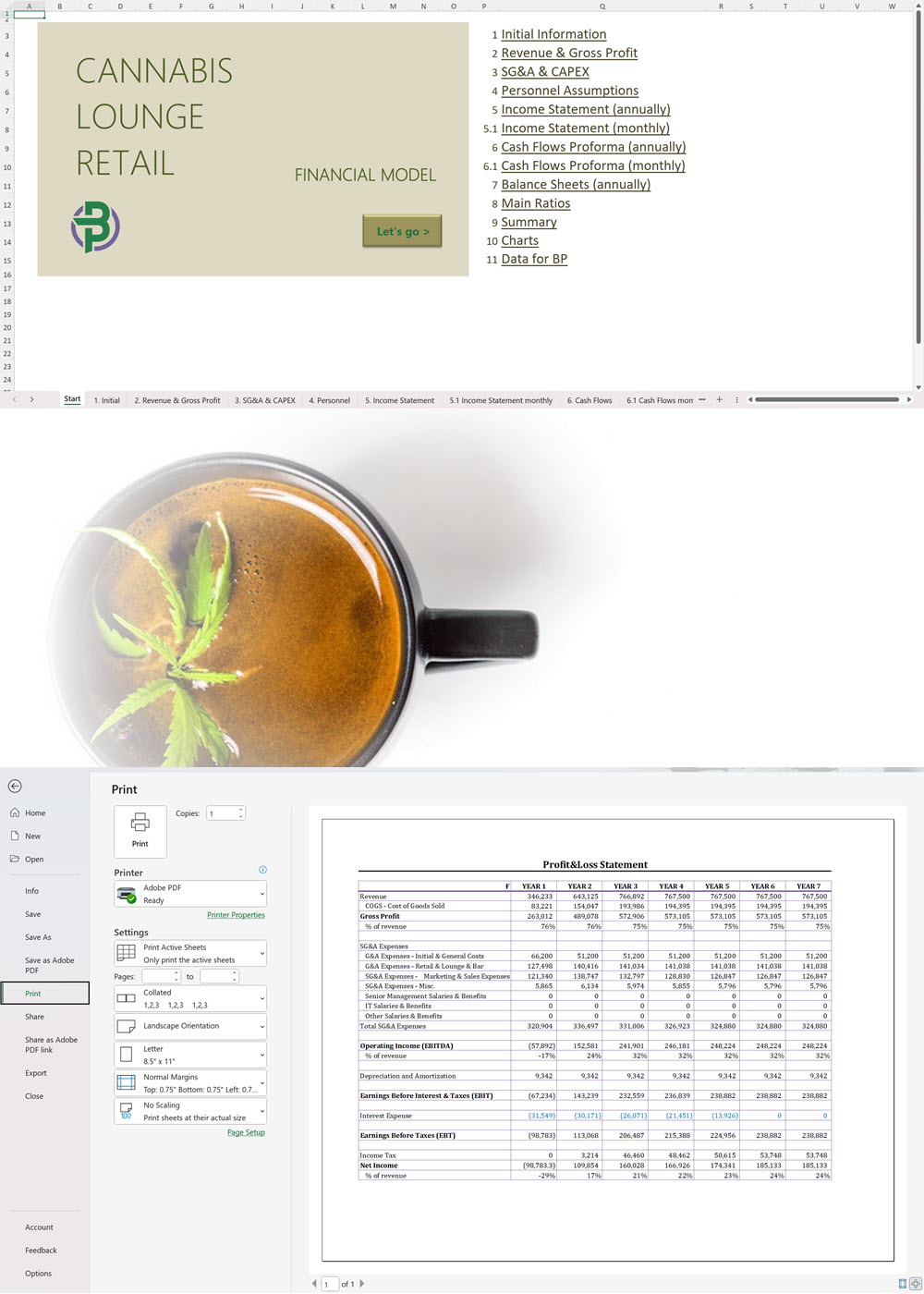
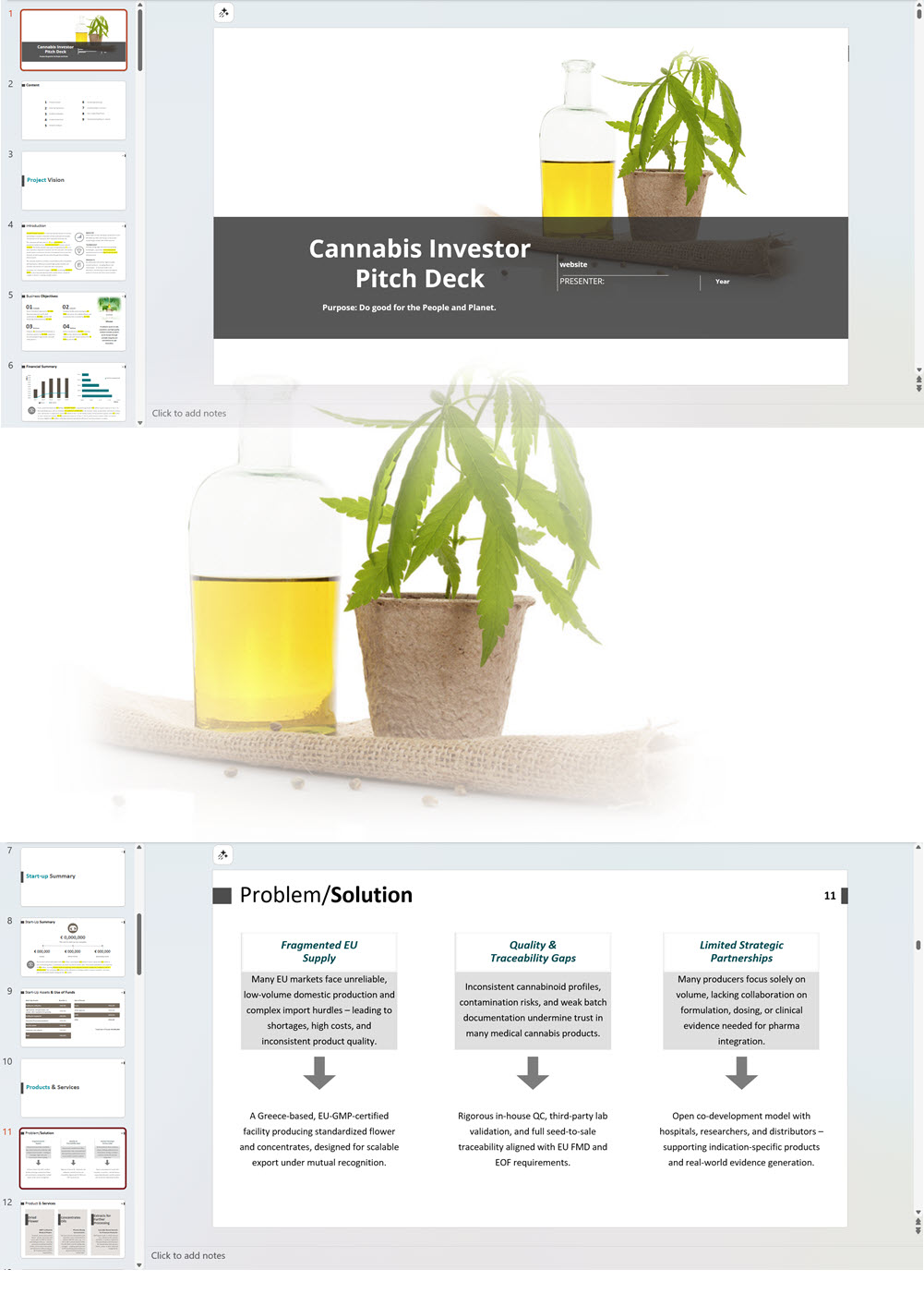
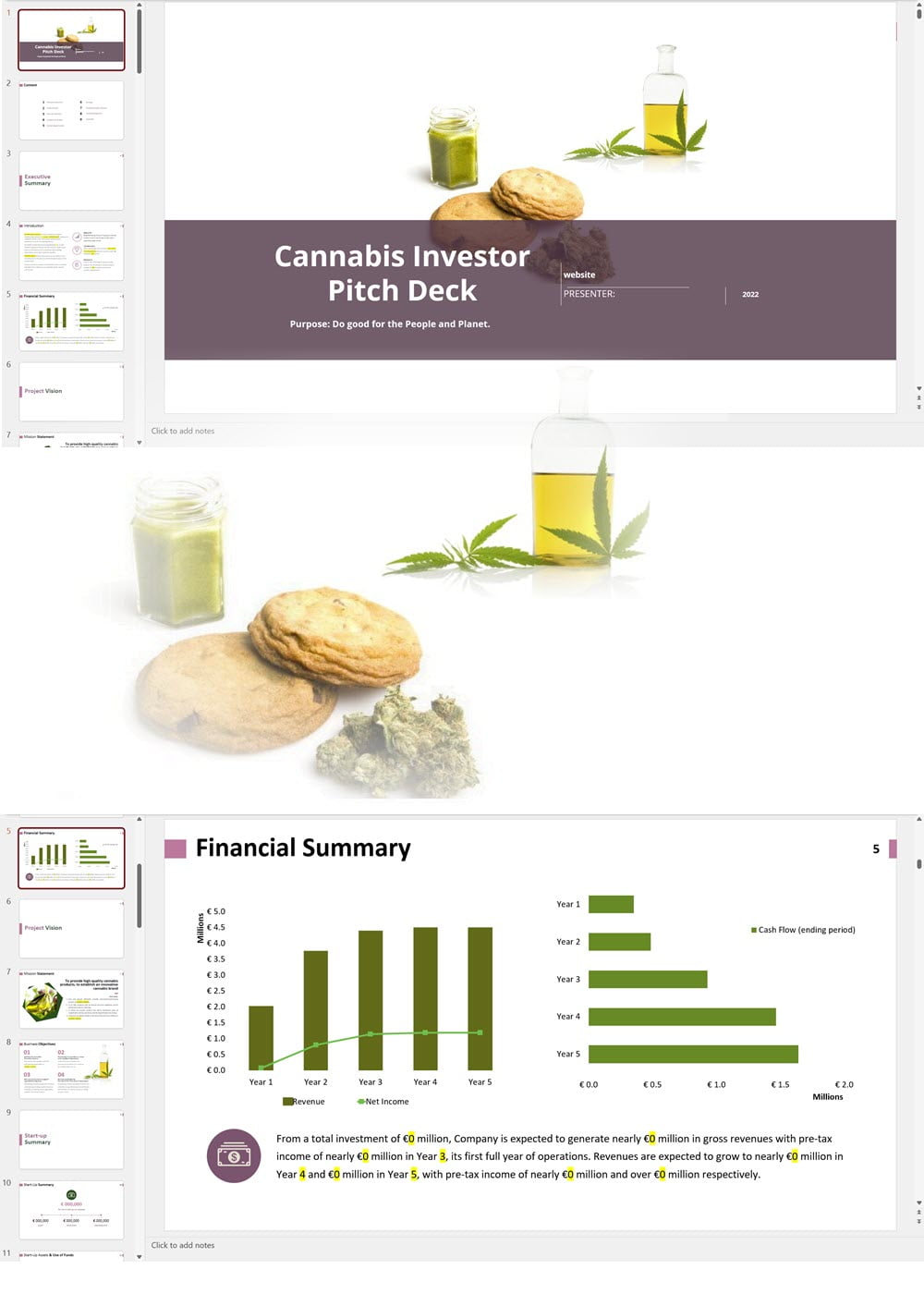
Main steps in short:
- Understand regulatory and legal requirements. Do market research.
- Decide the type of cannabis business. Choose a location. Check local zoning regulations.
- Develop a solid business plan. Secure financing.
- Submit an application. Pay fees. Prepare and submit the required documents.
- Register your business as an employer and a tax payer.
- Keep track of your ongoing compliance requirements.
- License renewal.
In 2015, Malta decriminalized cannabis. In 2018, Malta enacted the Production of Cannabis for Medicinal and Research Purposes Act which regulates the production of cannabis for medicinal and research purposes. Medical cannabis can be prescribed to patients suffering from chronic pain, spasticity in multiple sclerosis and side effects of chemotherapy. Patients can use non-smoking forms of cannabis-derived products once they obtain a card showing that they’ve been approved by Malta’s Superintendent of Public Health.
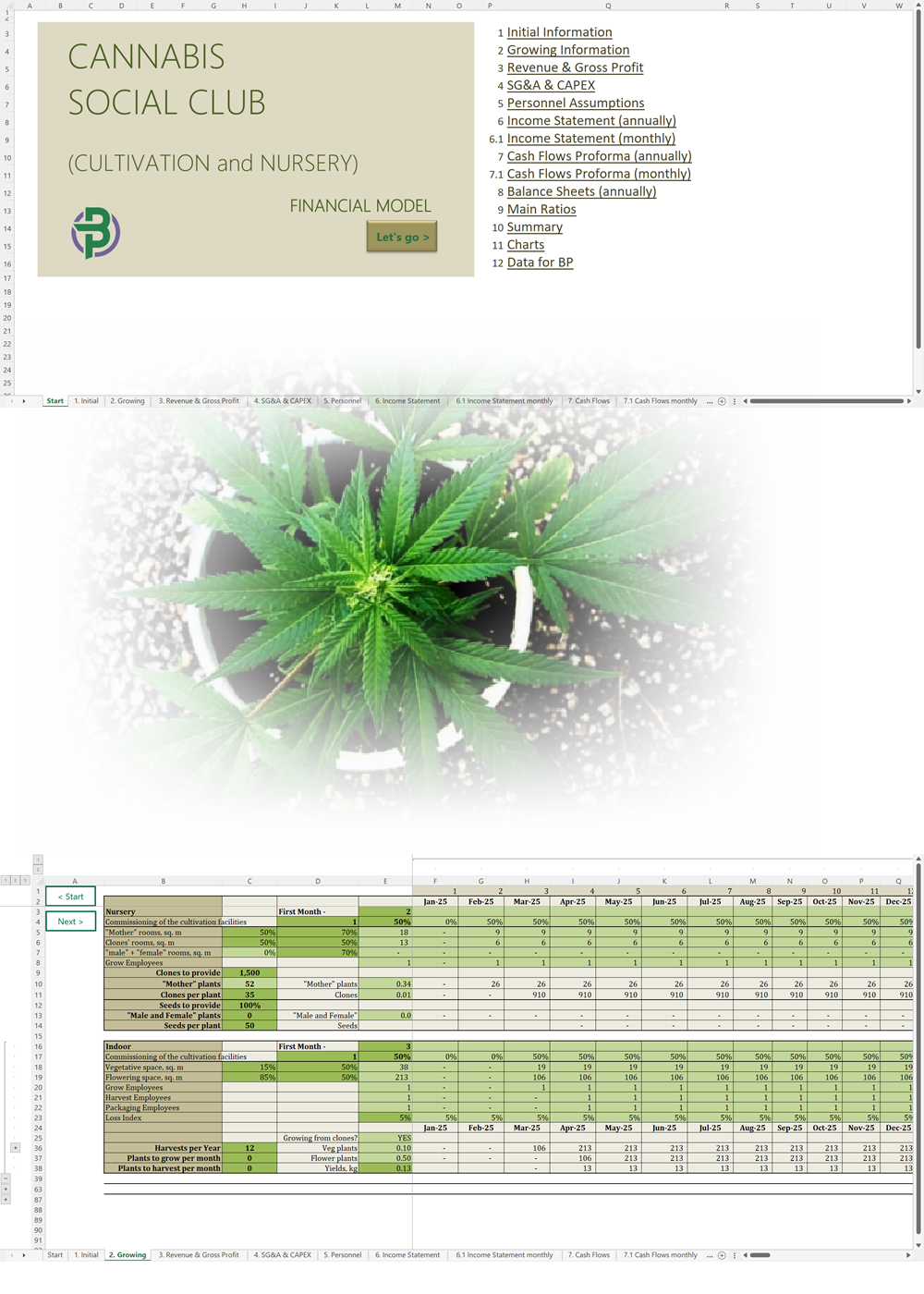
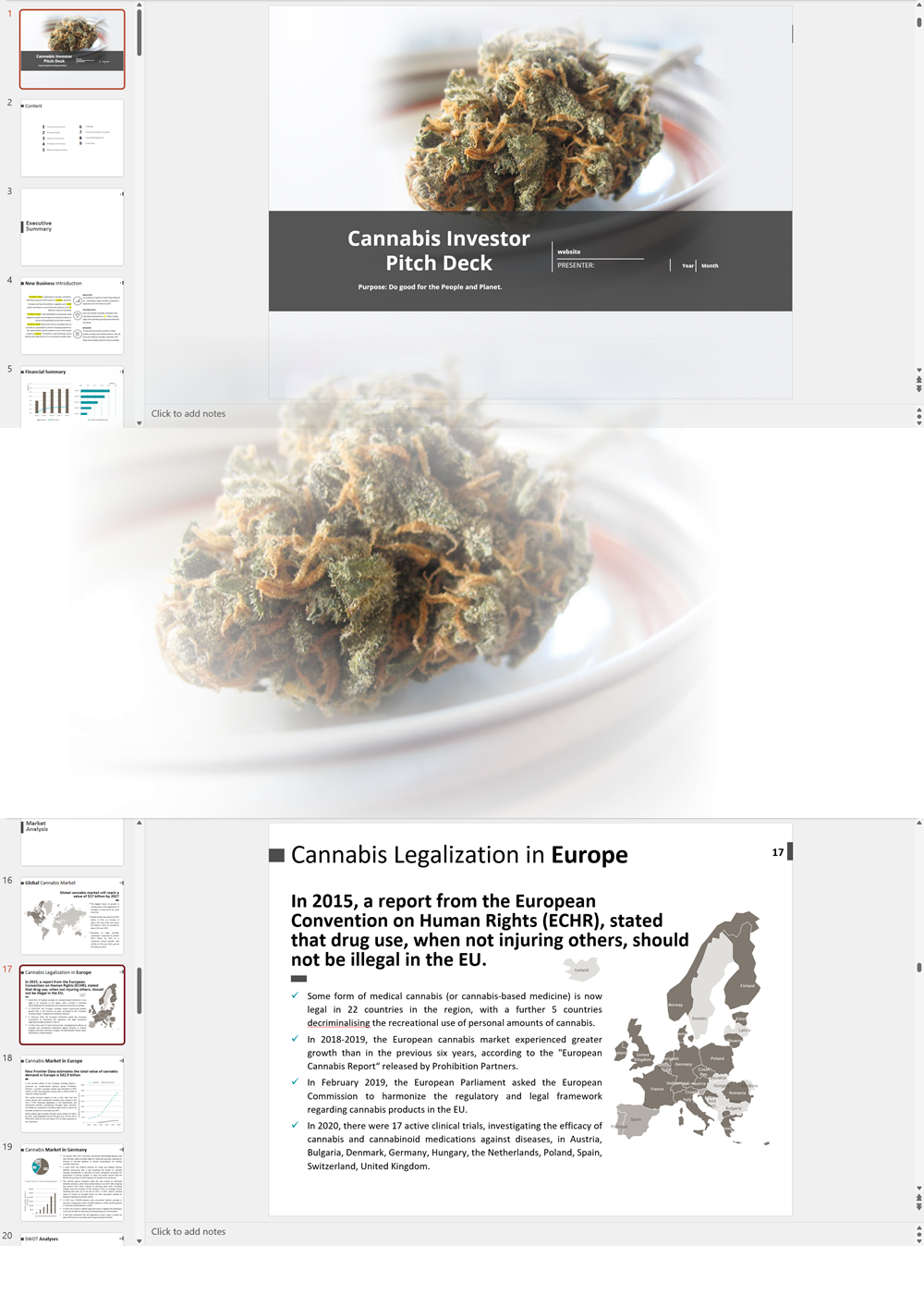
In December 2021, Malta became the first country in Europe to allow limited cultivation and possession of cannabis for personal use. Adults are allowed to carry up to seven grams of cannabis and grow no more than four plants at home. It also created cannabis social clubs – called Cannabis Harm Reduction Associations (CHRAs) – which are non-profit cooperatives that can grow cannabis for distribution to members. The Authority for the Responsible Use of Cannabis (ARUC) is responsible for the regulation of CHRA licensees.
The CHRA license is issued by the ARUC. Licenses for activities related to medical cannabis are issued by the Superintendent of Public Health.
A Cannabis Harm Reduction Association (CHRA) is a non-profit organization that is allowed to grow cannabis and distribute it to only its members. A CHRA can have no more than 500 members and all members must be at least 18 years old. A CHRA is allowed to distribute cannabis in flower form or to produce and distribute cannabis resin, provided that it is produced exclusively from the same quality-controlled dried flowers cultivated by the licensed CHRA and that its production is limited to solventless extraction methods only.
A CHRA is allowed to charge admission and membership fees or fees relating to the consumption of cannabis by its members. It is also allowed to generate income from the distribution of cannabis cultivated by the association and permitted goods to its members.
Application fee: €1,000
Annual license fees depend on the category of CHRA:
| Number of members | Annual License Fee | |
| Category 1 | Up to 50 | €1,000 |
| Category 2 | From 51 to 100 | €5,000 |
| Category 3 | From 101 to 250 | €13,000 |
| Category 4 | From 251 to 350 | €18,000 |
| Category 5 | From 351 to 500 | €26,000 |
CHRAs will also have to pay to the ARUC:
- A harm reduction contribution equivalent to 5% of the income that is generated by the CHRA.
- A community projects contribution equivalent to 10% of the amount of retained earnings recorded on the CHRA’s financial statement with respect to each financial year.
This license allows to manufacture cannabis-based products for medicinal purposes. There is an option to also cultivate cannabis.
Application/Renewal fee: €25,000
If the manufacturing includes local cultivation, the following additional contribution per footprint allocated for cultivation applies:
- €5 per m2 up to 2000m2
- €20 per m2 for part of footprint exceeding 2000m2 up to 4000m2
- €40 per m2 for part of footprint exceeding 4000m2 up to 6000m2
- €100 per m2 for part of footprint exceeding 6000m2
Annual fee: €25,000
This licence allows to carry out research activities related to cannabis.
Application/Renewal fee: €10,000
Annual fee: €8,000
This licence allows to source to Malta cannabis-based products or synthetic cannabinoid products licensed under the Medicines Act or manufactured under EU Good Manufacturing Practice.
Fees are for each form and strength:
Application/Renewal fee: €450
Fast track application fee (15 working days): €3,000
Annual maintenance fee: €275
- Develop a solid business plan. Secure financing.
- Submit an application. Pay fees. Prepare and submit the required documents.
CHRA licensing process:
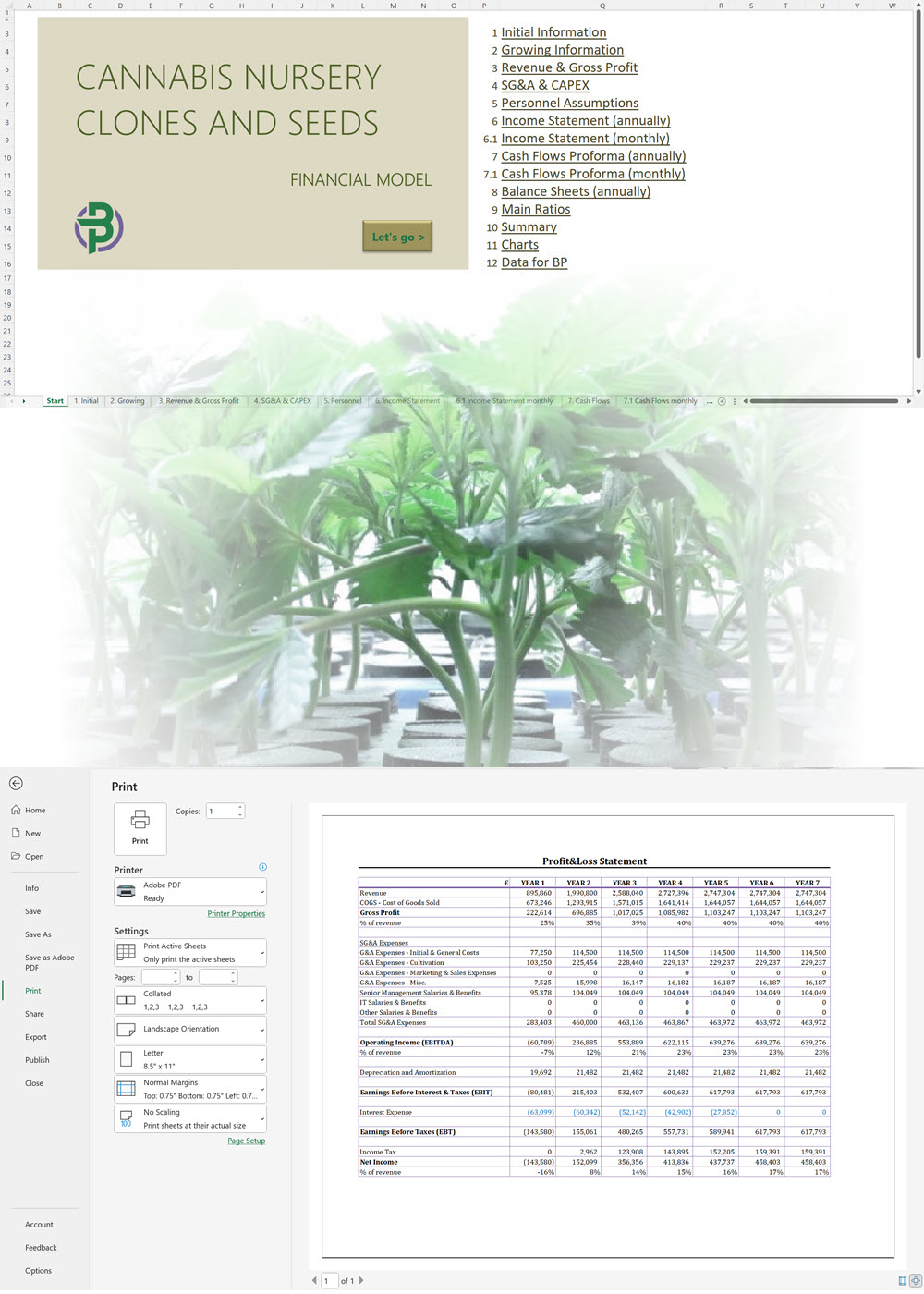
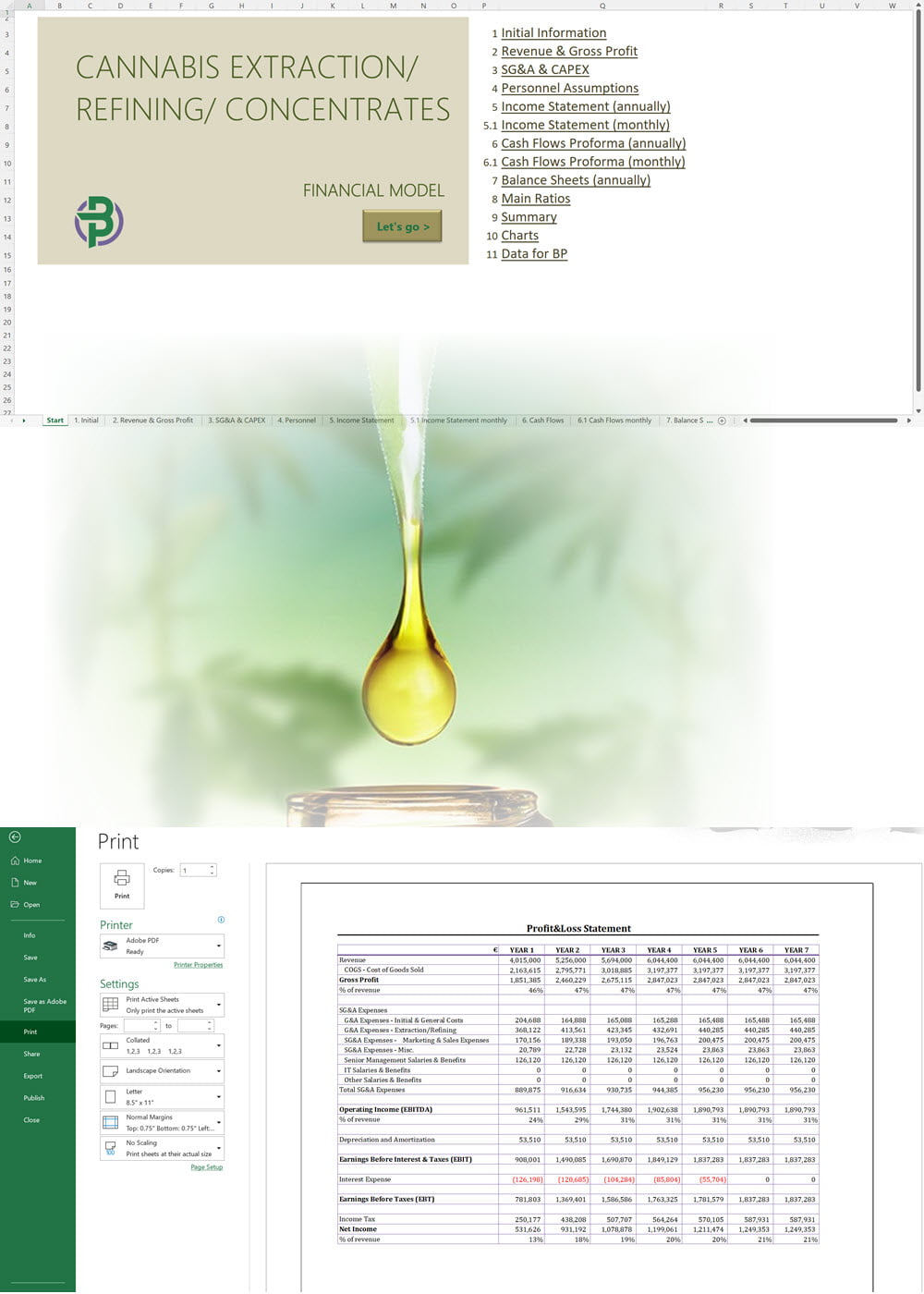
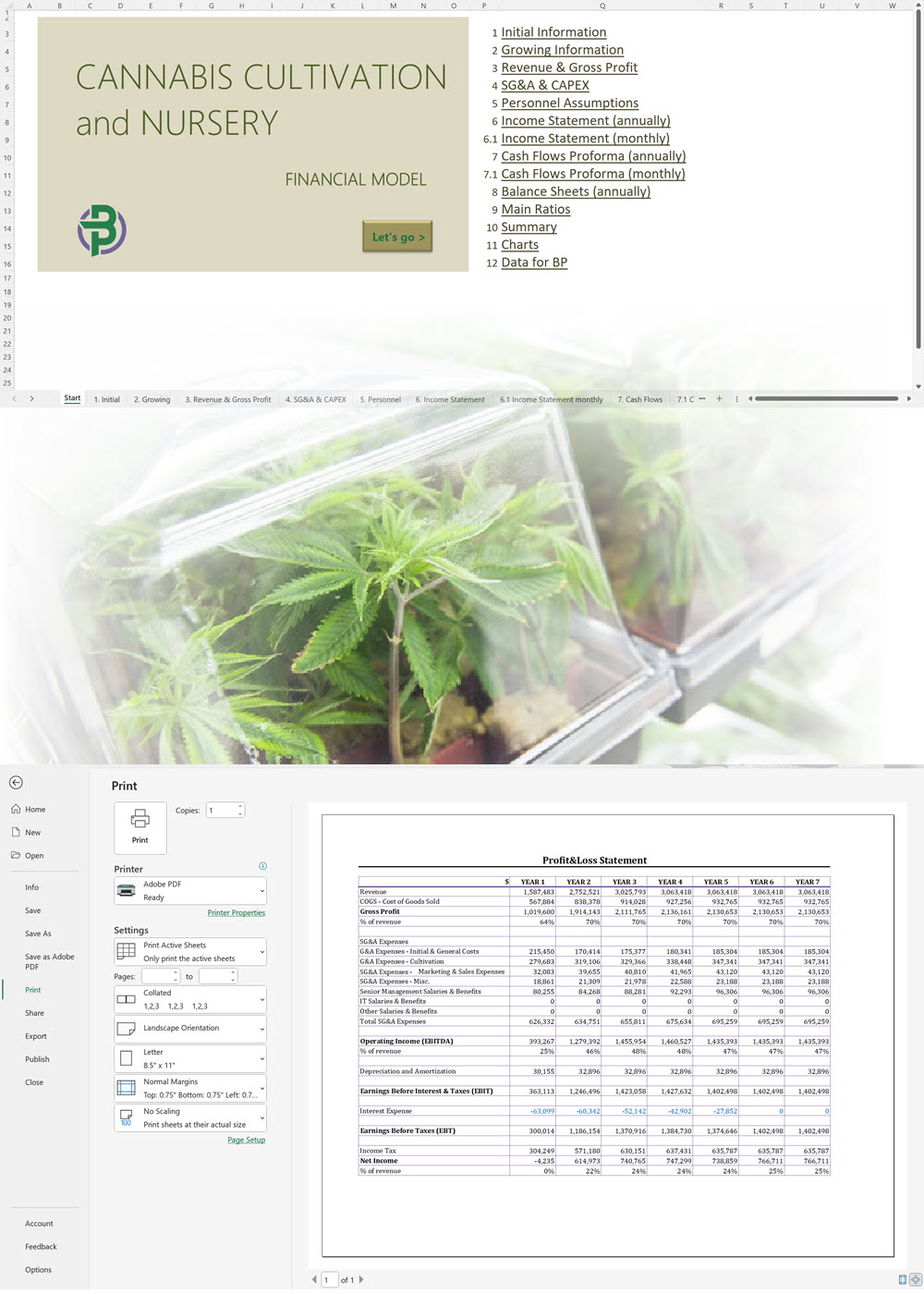
The applicant must submit the proposed name for the CHRA, its registered address, contact number and email. For each one of the founding members, administrators and Key Persons, the following information must be provided: The applicant must insert the addresses of the sites from where they plan to operate. The applicant shall provide the following documentation: An application for the CHRA license requires a three-year financial forecast with projected financial statements. Presenting this is made simple with our Financial Model Templates. All you need to do is input your most important assumptions and the model will create a set of projections, including ready-to-print financial statements, instantly! We offer a wide range of Excel cannabis financial models customized for activities ranging from cultivation to distribution. Input key assumptions and view the impact on your business' financial forecasts right away! Licensing process for activities relating to medical cannabis: To manufacture cannabis-based products for medicinal and/or research purposes, the applicant must obtain a Letter of Intent (LOI) from Malta Enterprise, a License for the Production of Cannabis for Medicinal and Research Purposes, an EU-Good Manufacturing Practice (EU-GMP) certificate and a Facility Security Clearance Certificate. Information that must be submitted with the application for the License for the Production of Cannabis for Medicinal and Research Purposes is not limited to: To trade in cannabis-based products for medicinal use, a wholesale dealer’s license under the Medicines Act and a Notification of Approval are required. The Notification of Approval is a product-specific notification issued by the Superintendent of Public Health, certifying the authorization for licensed wholesale distributors to source finished EU-batch released cannabis-based medicinal products and place them on the market in Malta. Licensees are also required to comply with the relevant sections of the European Union Good Distribution Practice (EU-GDP) Guidelines published by the European Commission. Information that must be submitted with the application for a wholesale dealer’s license is not limited to: Before the license is issued, the premises will be inspected by the Superintendent of Public Health. The Medicines Authority shall establish the period for which the license is valid. Information that must be submitted with the application for wholesale distributors to place cannabis-based products or synthetic cannabinoid products on the market is not limited to: A full list of all the information and documents that must be attached to the applications can be found in the application forms provided by the Medicines Authority. Whether you are applying for a CHRA or a medical cannabis license, you may need to secure finance for starting your business and this will require a professional business plan. Our Business Plan Templates will help you to create a business plan that effectively conveys your business goals and strategies, market analysis, operating plan, organizational structure and financial information. We offer a wide range of '70% ready-to-go' cannabis business plan templates that will help you prepare a professional cannabis business plan document supported with expert financials and an investor pitch deck. All our templates are completely open and editable and you can use Word, Excel and PowerPoint to work with them. All licensees must comply with the regulations regarding: Licensees involved with medical cannabis-based product manufacturing must ensure ongoing compliance with EU-GMP standards. Licensees involved with the wholesale or import of cannabis-based products or synthetic cannabinoid products must ensure ongoing compliance with EU-GDP standards. All licenses for activities relating to medical cannabis are valid for a period of 3 years. To renew them, an application must be submitted using the same form as for the initial license application and the relevant renewal fee must be paid. The CHRA license is valid for a period of 1 year. To renew the license, an application must be submitted using the same form as for the initial license application and the relevant renewal fee must be paid. CHRAs may have a sign affixed at the entrance of the distribution site and at the registered address to indicate that the site is used by the CHRA. CHRAs may request the ARUC’s consent to have their own website where content may be accessed only through login credentials secured through two-factor authentication; these credentials may only be assigned to registered members. However, the use of techniques which artificially improve the visibility of the website in search algorithms is prohibited. CHRAs may also have their own social media pages and other digital channels as long as the content is set to “private” and is visible to registered members only. Advertisement of medical cannabis to the public is prohibited. Licensees may only advertise medical cannabis to persons who are qualified to prescribe it. Our cannabis financial models and cannabis business plan templates will help you estimate how much it costs to start and operate your own cannabis business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates. Cannabis business plan templates for other countries are available at cannabusinessplans.com.Plug-and-Play Financial Model Templates
'70% ready to go' business plan templates
Cannabis Advertising Regulations
Cannabis Social Club Business Plan Sample, Malta
'70% ready to go' business plan templates
Best Selling Templates

Cannabis Cultivation Business Plan Template
Price range: €75 through €350
Select options
This product has multiple variants. The options may be chosen on the product page 
Cannabis Dispensary Investor Pitch Deck Template
€75
Select options
This product has multiple variants. The options may be chosen on the product page 
Cannabis Financial Model All in One
€250
Select options
This product has multiple variants. The options may be chosen on the product page