Medical Cannabis
The medical cannabis pilot program in Denmark began in January 2018 and is currently serving around 500 unique patients per quarter. The program allows physicians to prescribe medical cannabis from a list of five flower and full-spectrum oil products for the following conditions: painful spasms due to multiple sclerosis or spinal cord injury, nausea after chemotherapy, neuropathic pain, although some leeway remains depending on other conditions. 100% of the costs of patients participating in the trial scheme if their condition is terminal are covered by the country’s national insurance agency, and coverage of 50% of the cost is provided in other cases up to DKK 10,000 per year. Patients may also choose to obtain magistral cannabinoid isolates outside of the pilot scheme without reimbursement.
The medical cannabis products allowed for use in the trial can be found on this page.
The scheme was originally conceived as a four-year project, but in May 2021 a majority of Parliament voted to extend the project for another four years, with it now running until at least 2025.
On November 19, 2024, the government proposed that the medical cannabis prescription system should be made permanent after an evaluation of the pilot program showed that medical cannabis was benefitting many patients. Representatives from other political groups have been invited to discuss this proposal.
There are currently four ways that patients can access medicinal cannabis in Denmark:
- Through the pilot program.
- As an approved medicine. There are currently two approved medicines: Sativex and Epidyolex.
- Medicines Marinol and Nabilone, which contain synthetically produced cannabinoids, prescribed to patients by a doctor with a special dispensing permit. These medicines can be imported if the Danish Medicines Agency grants a Danish doctor permission to prescribe it to specific patients.
- Medicinal products such as capsules or oils containing ingredients form cannabis prepared in a pharmacy for a specific patient’s needs according to a prescription from a doctor.
The medical cannabis pilot program evaluation report proposed that all four schemes should be streamlined and standardized and the licensing process for new products should be simplified.
Medical Cannabis Authorizations in Denmark
It is important for the applicant to understand the following terms before choosing a permit:
A cannabis end product is a finished cannabis product that a doctor can prescribe to a patient and which a pharmacy manufactures and dispenses to the patient.
A cannabis intermediate product is used by a pharmacy to manufacture the final cannabis product.
A cannabis starting product is used by an intermediate product manufacturer to produce the cannabis intermediate product.
The following medical cannabis authorizations are available in Denmark:
Allows to produce cannabis starting products with cultivation activity as well as manufacture cannabis bulk and cannabis precursor products. Cannabis bulk is a cannabis-based product intended for the purpose of further processing. Cannabis precursor is a cannabis-based product that is in finished product form and in consumer-ready packaging.
Application fee: DKK 145,579
Annual fee: DKK 59,889
Allows to produce cannabis starting products without cultivation activity as well as manufacture cannabis bulk and cannabis precursor products. Cannabis bulk is a cannabis-based product intended for the purpose of further processing. Cannabis precursor is a cannabis-based product that is in finished product form and in consumer-ready packaging.
Application fee: DKK 139,982
Annual fee: DKK 46,455
Allows the production of cannabis intermediates.
Application fee: DKK 78,691
Annual fee: DKK 46,455
Allows the cultivation and handling of cannabis for the purpose of developing cannabis for medical use.
Application fee: DKK 48,813
Annual fee: DKK 14,864
Manufacturing Cannabis Products for Use in the Trial Scheme
Licensing process:
- Submit a complete application with all the relevant documents to the Danish Medicines Agency (DMA).
- An inspection will follow, performed by the DMA.
- If the inspection gives satisfactory results, the DMA will issue a certificate authorizing the company to perform the requested activities with medical cannabis and it also provides proof that the company complies with Good Manufacturing Practice for medicines.
Information about the following is required to be included with the application:
- Activities that will be performed with medical cannabis.
- Contractors for outsourced activities, if applicable.
- The Expert Person of the company.
- The person responsible for security.
- The company’s owner or director.
- Address of the proposed location for the medical cannabis establishment.
More information on the application process can be found in the following guides:
Special Development Scheme
On January 1, 2018, Denmark introduced a special development scheme under which companies can apply for an authorization from the DMA to cultivate and handle cannabis for the purpose of producing medical cannabis. This scheme runs in parallel with the medicinal cannabis pilot program. Though the authorization does allow to grow and develop medical cannabis, it does not allow the company to supply medical cannabis products for patient use in Denmark or export them; for this, a cannabis bulk manufacturing permit is required. The DMA official website states that the purpose of an authorization under the development scheme is to give companies “an opportunity to build competencies and develop methods and products within medical cannabis for use in the trial scheme”. Later, the company can apply for a cannabis bulk manufacturing permit in order to be able to supply its products for patient use and export.
To obtain the authorization, the applicant must submit a complete application and a project description to the DMA and the DMA must also carry out an inspection. The project description should demonstrate how the applicant’s proposed project will contribute to the development of cannabis products that can be included in the medical cannabis trial scheme or used in pharmaceutical production.
The application must contain the following information:
- The type of activity the applicant wants to perform relating to medical cannabis. Possible activities include import, export, receipt of cannabis, cultivation, possession and preparation of products and solutions.
- The addresses of the locations where cultivation and/or processing will take place.
- If the applicant intends to cultivate cannabis, the cultivation area must be stated in hectares.
- Information about the person responsible for security and about the company’s owner or director. Both must sign their consent that the DMA can access information about personal circumstances from the National Police.
The project description must provide information on the following topics:
- Quality assurance.
- Personnel and training.
- Buildings and facilities.
- Equipment.
- Seeds and propagating material.
- Cultivation process.
- Harvesting process.
- Primary processing which includes washing, trimming and drying.
- Packaging of the harvested cannabis.
- Storage of cannabis product.
- Timetable for cultivation, harvesting and primary processing as well as when cannabis products will be included in the medical cannabis trial scheme.
- Destruction of cannabis procedure.
- Special safety measures relating to the cultivation and processing of cannabis.
- Production of medicinal cannabis.
The DMA has a guide on how to apply for the authorization as well as a detailed guide on what information the project description should contain.
Inclusion of Medical Cannabis Products in the Trial Scheme
Companies wishing for their medical cannabis products to be included in the DMA’s list of products that can be used in the trial scheme, should submit an application to the DMA. Products can either be imported into Denmark or manufactured in Denmark using Danish-grown cannabis. The application forms and instructions on how to submit them can be found on DMA’s official website.
Application fee: DKK 55,176
Application processing times for inclusion of cannabis intermediates and related imported cannabis starting products:
Validation – 14 days
Assessment period – 50 days
Application fee: DKK 141,371
Application processing times for inclusion of cannabis intermediates and related starting products manufactured in Denmark:
Validation – 14 days
Assessment period – 70 days
Once the product is approved, the company must report information about prices, packaging and delivery capacity.
Cannabis Market in Denmark
According to a 2024 Evaluation Report, in 2021, there were 9 holders of a permit to produce bulk cannabis and cannabis derivatives and 11 holders of a permit to manufacture cannabis intermediates. After a few increases, the number of active permits to produce bulk cannabis and cannabis derivatives remained at 9 in November 2023 while the number of active permits to manufacture cannabis intermediates fell to 8. Permits issued under the special development scheme experienced a downward trend from 2020 to 2023, falling from 44 in the second half of 2020 to 20 in November 2023.
As of January 2025, there are 11 registered medical cannabis companies under the development scheme, 8 registered medical cannabis companies producing bulk cannabis and cannabis derivatives and 9 registered medical cannabis companies manufacturing medical cannabis intermediates.
There were nearly 3,000 patients in 2018. In 2019, patient numbers reached 3,569 and then dropped back to just below 3,000 in 2020. From there, patient numbers began to fall with 2,494 patients registered in 2022. In 2021 and 2022, patients acquiring medical cannabis through magistral medicines accounted for around 63-64% of all patients using medical cannabis. The next largest group are patients acquiring medical cannabis through the trial program.
Prescriptions, however, did not decline with patient numbers. They initially rose by 33.5% from 2018 to 2019. In the next years they continued to grow, reaching 13,432 prescriptions in 2022.
From 2020 to 2023, 20,000 prescriptions for medical cannabis have been issued to around 1,800 patients.
Products from Aurora’s facilities hit the German shelves in February 2021, marking the country’s first international export. In all of 2021, 3,424kg of medical cannabis were exported from Denmark. This rose by 55% to 5,313kg in 2022 and from January to November 2023, 4,501kg were exported.
In Denmark, the majority of the market share is in single substance extracts, both THC and CBD, with flowers accounting for only 12% of the total unlicensed market.
Denmark Cannabis Market Infographics
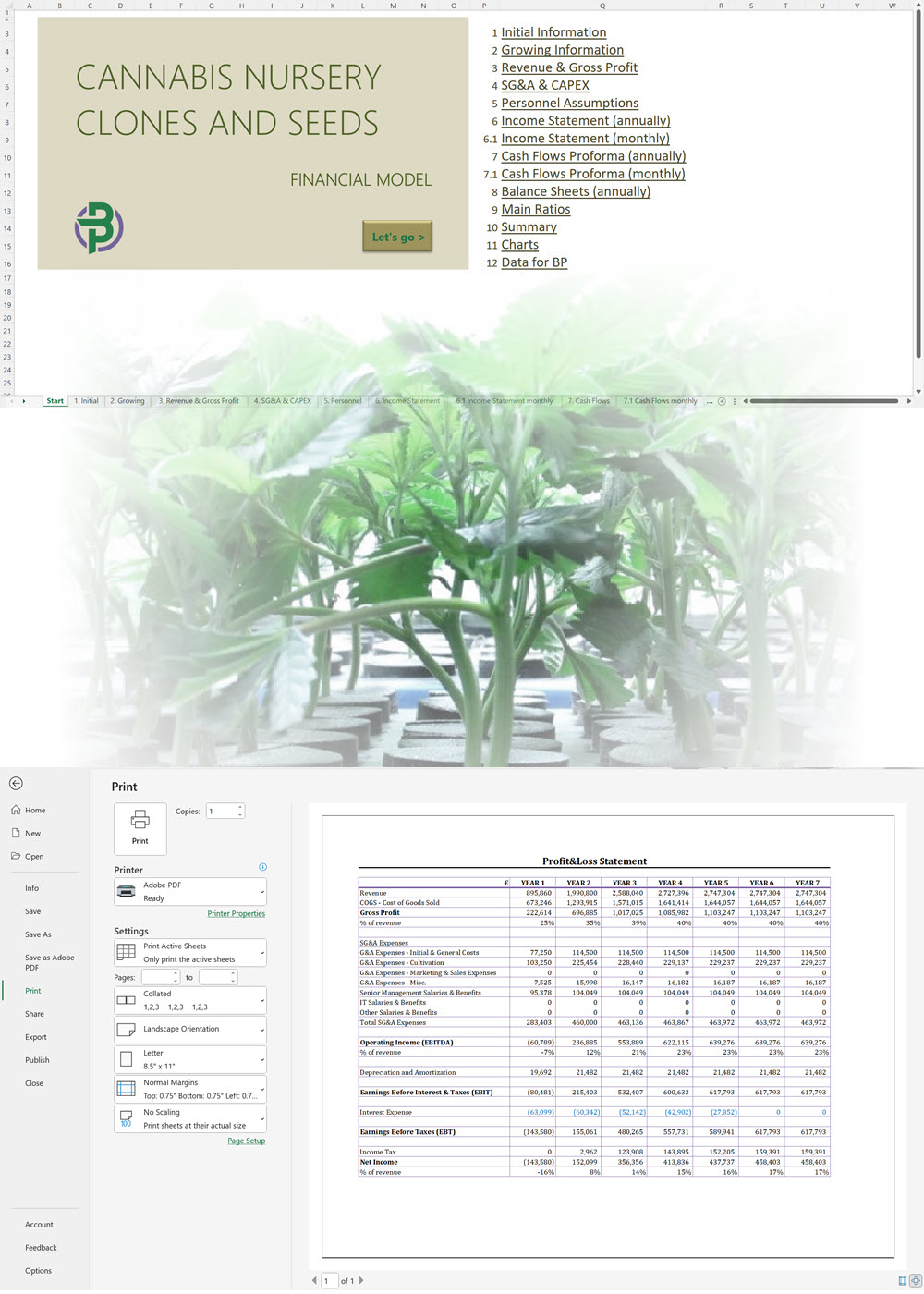
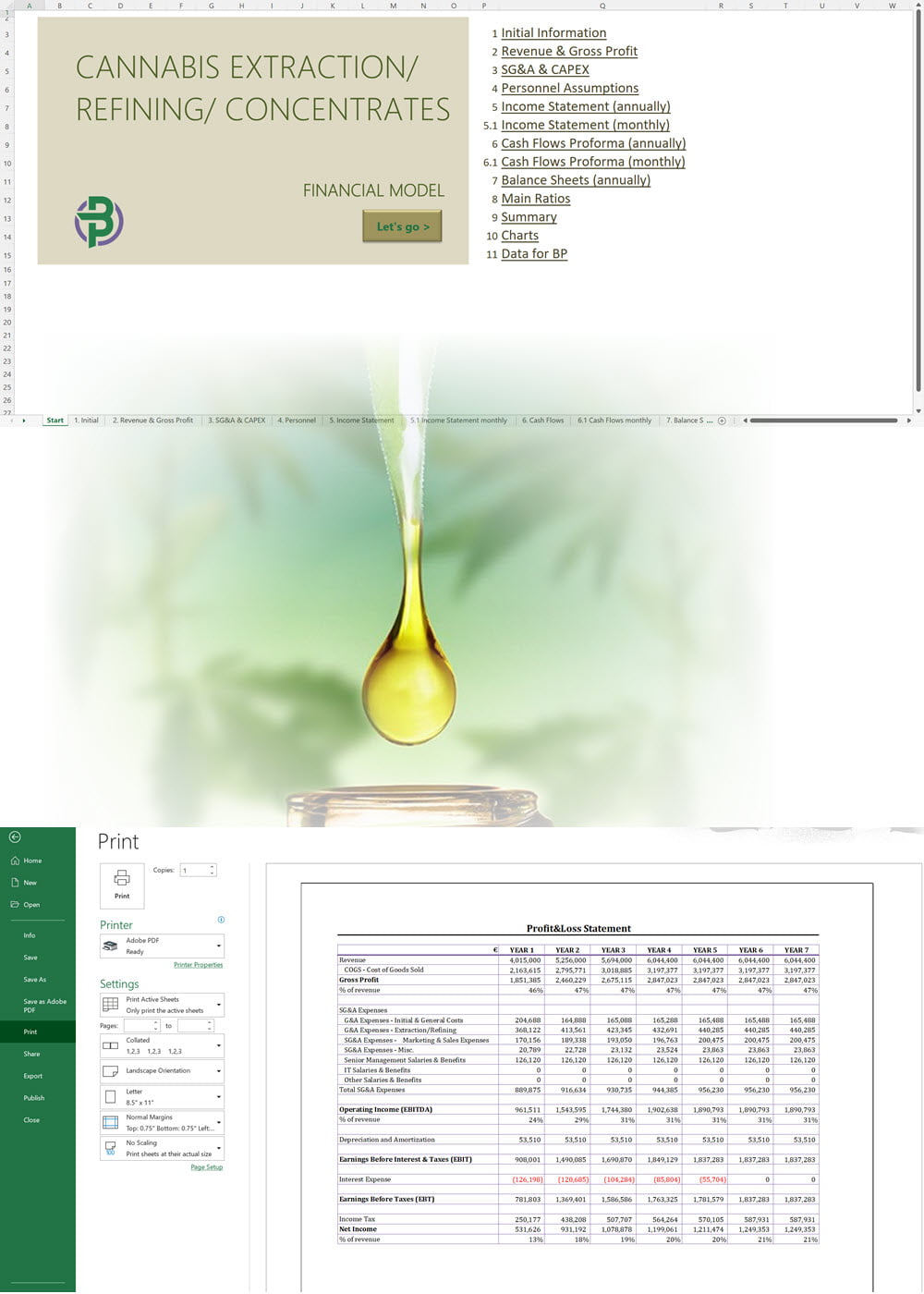
Cannabis Extraction/Concentrates Business Plan Sample, Denmark
'70% ready to go' business plan templates
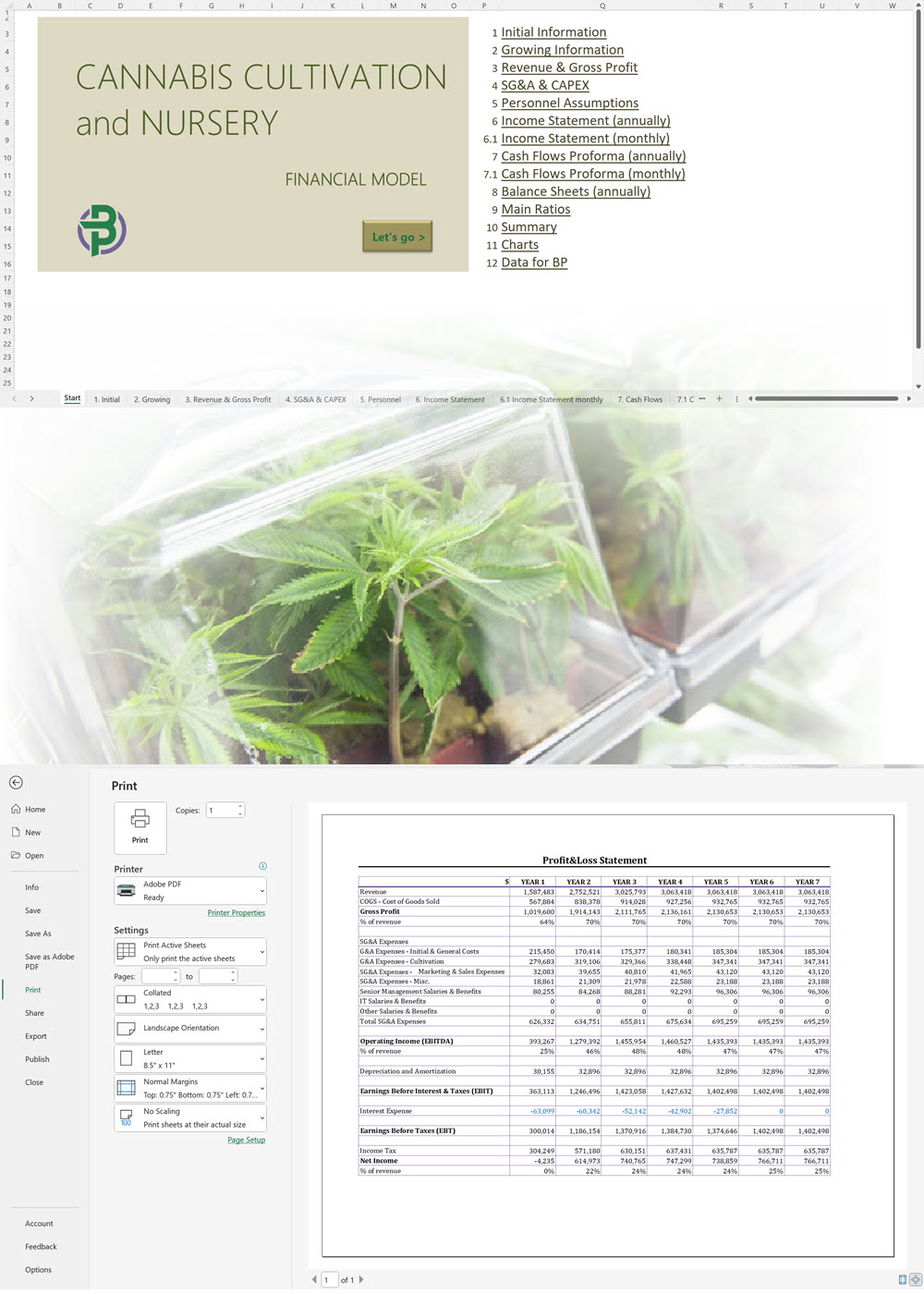
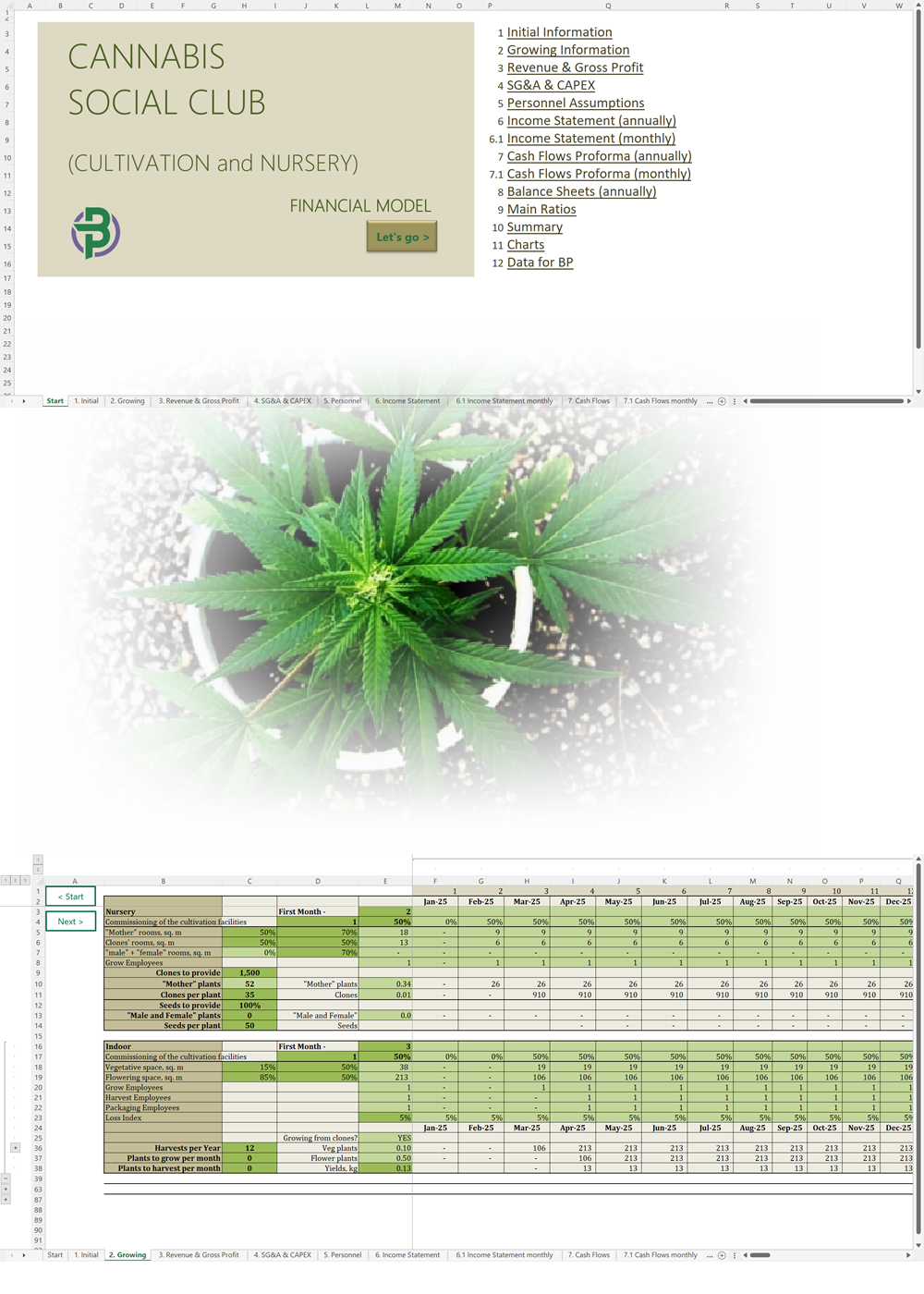
Our cannabis financial models and cannabis business plan templates will help you estimate how much it costs to start and operate your own cannabis business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates.
Best Selling Templates
-

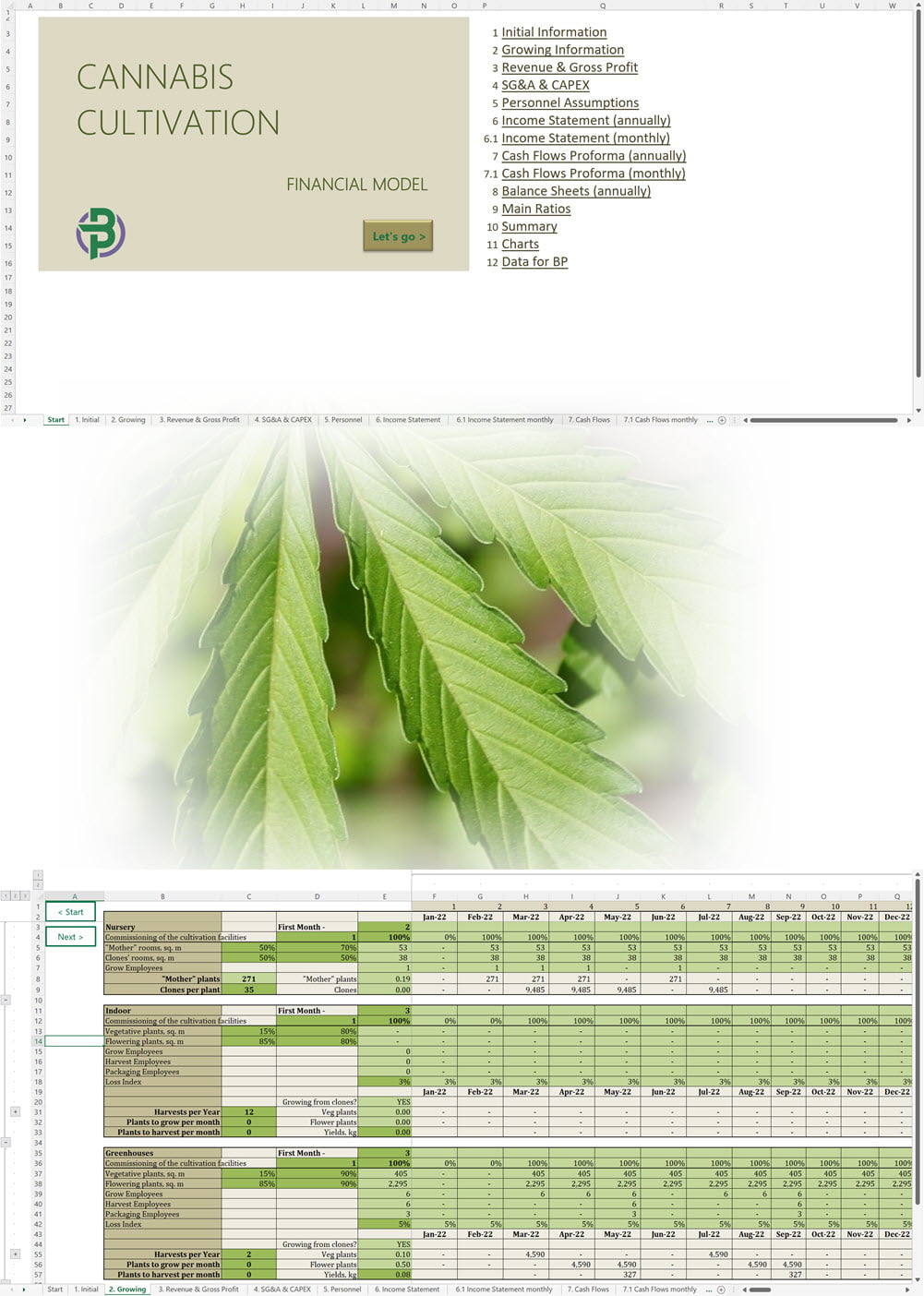
Cannabis Cultivation Business Plan Template
Price range: €75 through €350 Select options This product has multiple variants. The options may be chosen on the product page -

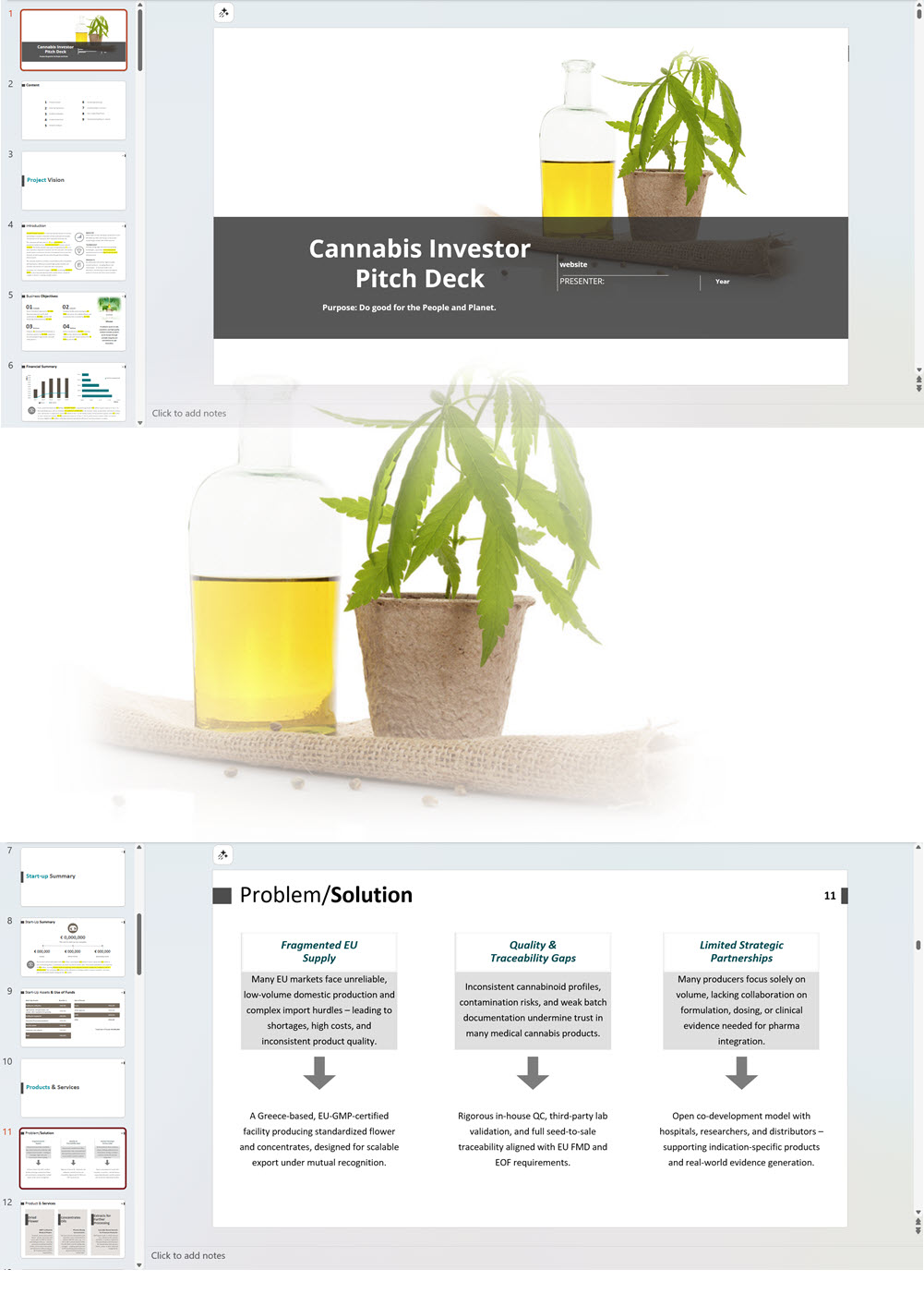
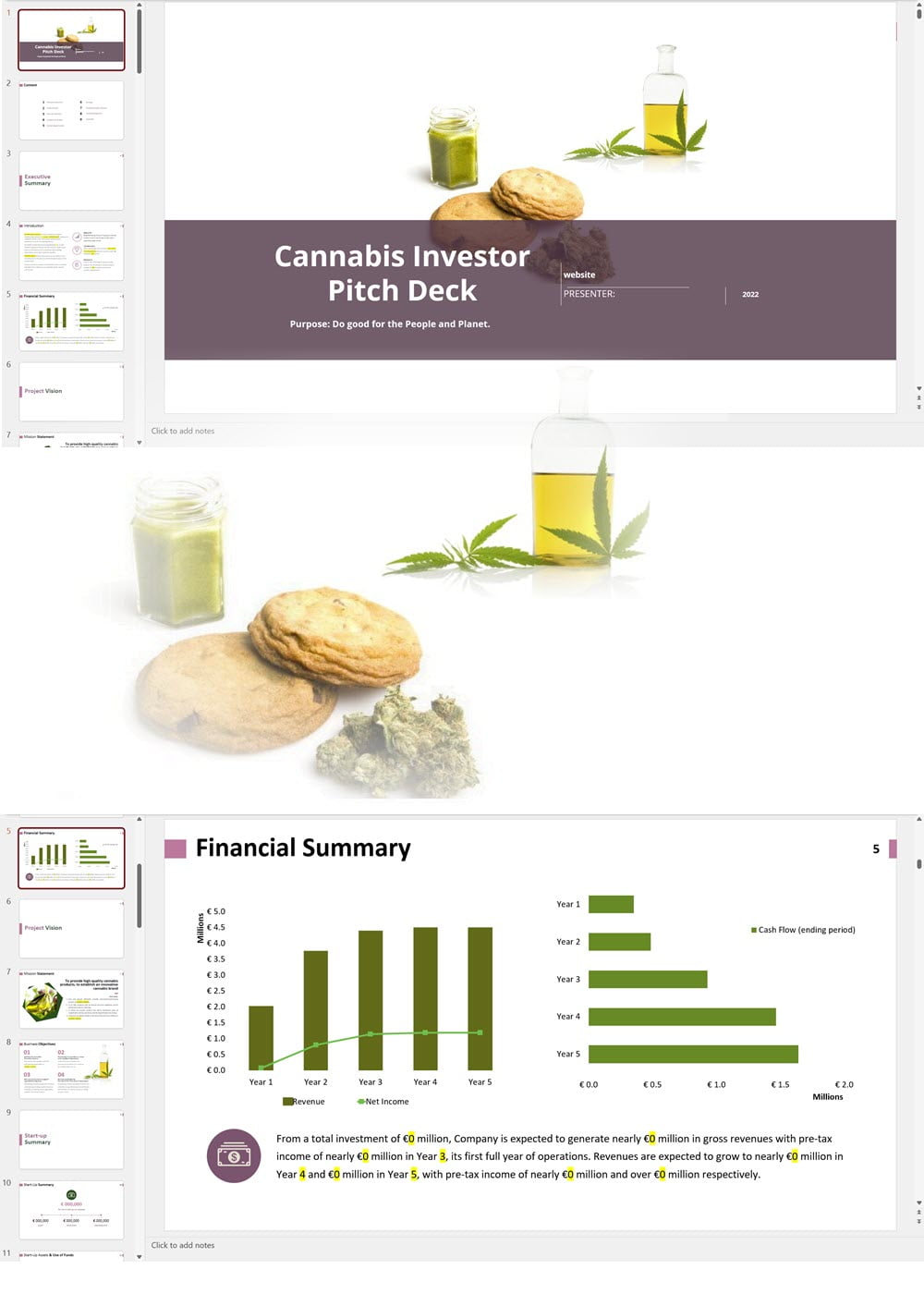
Cannabis Dispensary Investor Pitch Deck Template
€75 Select options This product has multiple variants. The options may be chosen on the product page -

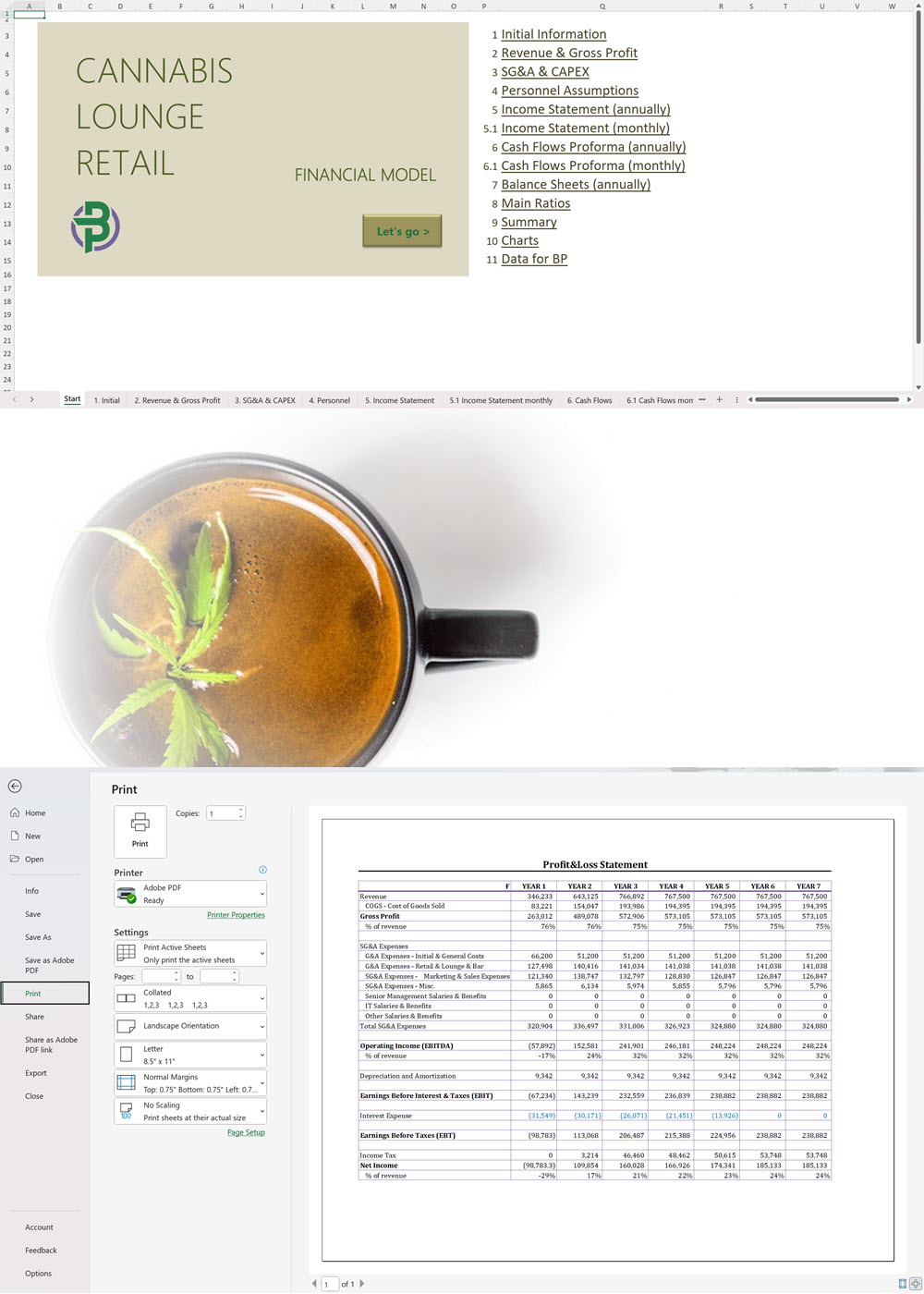
Cannabis Financial Model All in One
€250 Select options This product has multiple variants. The options may be chosen on the product page
We also offer hemp/CBD business plan templates at hempcbdbusinessplans.com.
Templates for other countries can be found at cannabusinessplans.com.