Main steps in short:
- Understand for what purpose you wish to export cannabis.
- Familiarize yourself with GACP regulations.
- Obtain the EU-GMP Certificate or a Written Confirmation.
- Obtain a marketing authorization for your medical cannabis product.
- Carry out market research. Identify specific EU countries to which you wish to export cannabis. Research cannabis regulations in those countries.
- Find your partners and acquire the necessary import and export permits.
Over the last few years, the cannabis market in Europe has been on a rise. A significant surge occurred in 2018-2019, when six new countries legalized medical cannabis. As of May 2025, some form of medical cannabis is legal in 22 European countries with an additional 11 countries having decriminalized the recreational use of personal amounts of cannabis and 3 countries have legalized recreational cannabis. The first country to legalize cannabis for recreational use was Malta who did so in December 2021. Luxembourg and Germany followed, legalizing recreational cannabis in June 2023 and February 2024, respectively. Due to its size and political importance, Germany’s legalization is expected to serve as an example and encourage other countries in the EU to also progress towards legalizing recreational cannabis.
While there has been progress towards greater adult-use cannabis legalization, the main market opportunity in Europe remains to be medical cannabis. Germany is the largest medical cannabis market in the region, with patient numbers growing rapidly since the implementation of the German Cannabis Act in 2024 – as of May 2025, it is estimated that there are nearly 900,000 medical cannabis patients in the country. Around 1 out of every 7 pharmacies can offer medical cannabis. Revenue from the medical cannabis industry in Germany was estimated to be around €420 million in 2024.
Currently, only cannabis for medical use can be imported into Europe. Many European countries that have legalized medical cannabis still rely heavily on imports to satisfy domestic demand. For instance, Germany’s imports of medical cannabis more than doubled from 32,503 kilograms in 2023 to over 72,000 kilograms in 2024. Canada is the largest importer, accounting for around 50% of Germany’s cannabis imports. Poland, the fifth most populous country in Europe, raised its medical cannabis import limit for 2024 from 6 tons to over 11 tons.
Reports by London-based advisory group Prohibition Partners value the European legal cannabis market at €354 million in 2022 and €516 million in 2023. Total cannabis sales are forecast to grow with a CAGR of 45% to reach €2.1 billion by 2027.
How to Start Exporting Medical Cannabis to Europe?
You may export cannabis flower cultivated at your facilities for the manufacture of cannabis medicines in Europe or you may choose to export finished medical cannabis products to Europe which can be accessed by patients – and these two activities would require compliance with different regulations and different certifications. You may find it helpful to hire a lawyer who is familiar with the European cannabis law and can assist you on your journey to exporting cannabis to Europe. Develop a solid cannabis business plan. Secure financing.
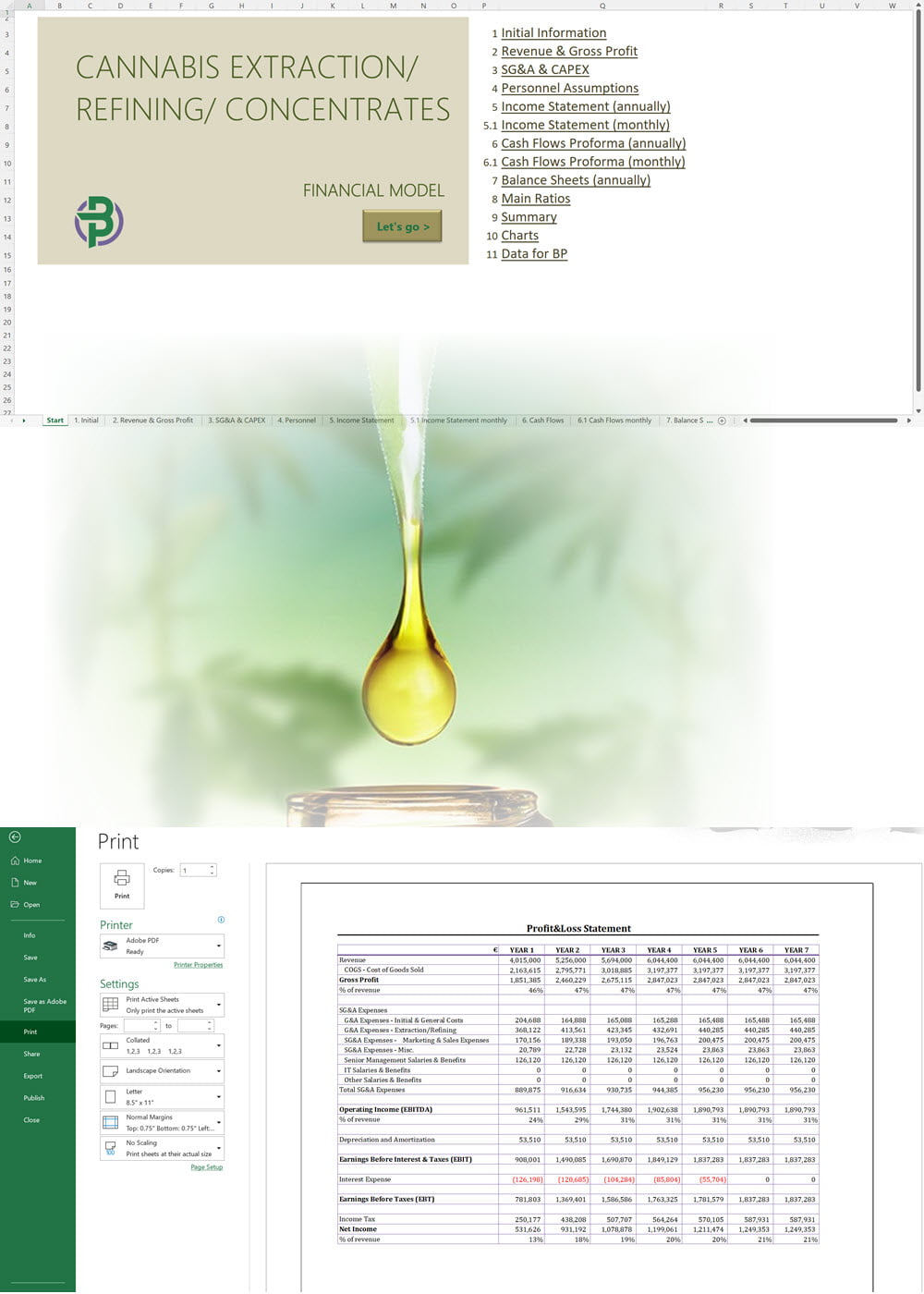
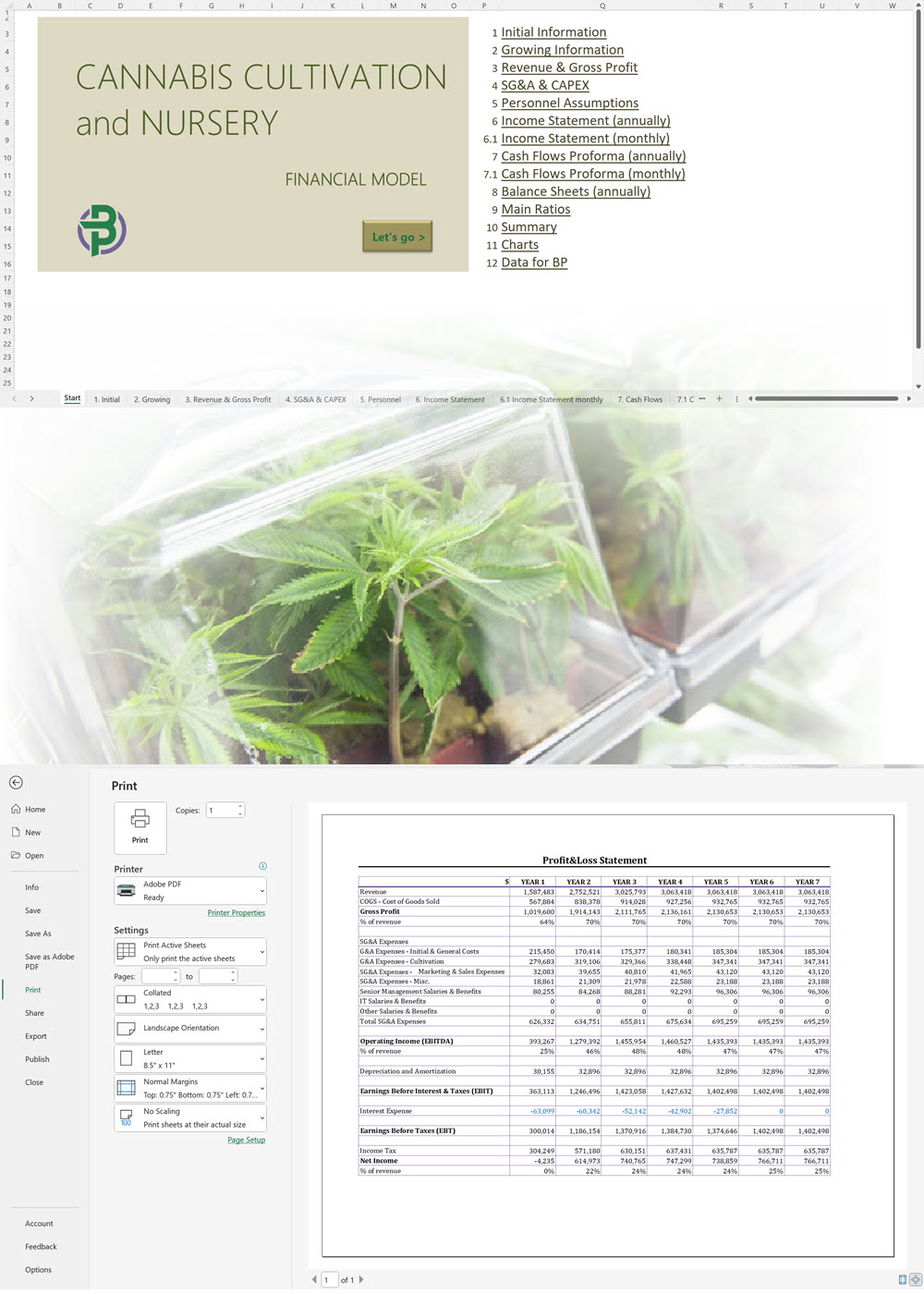
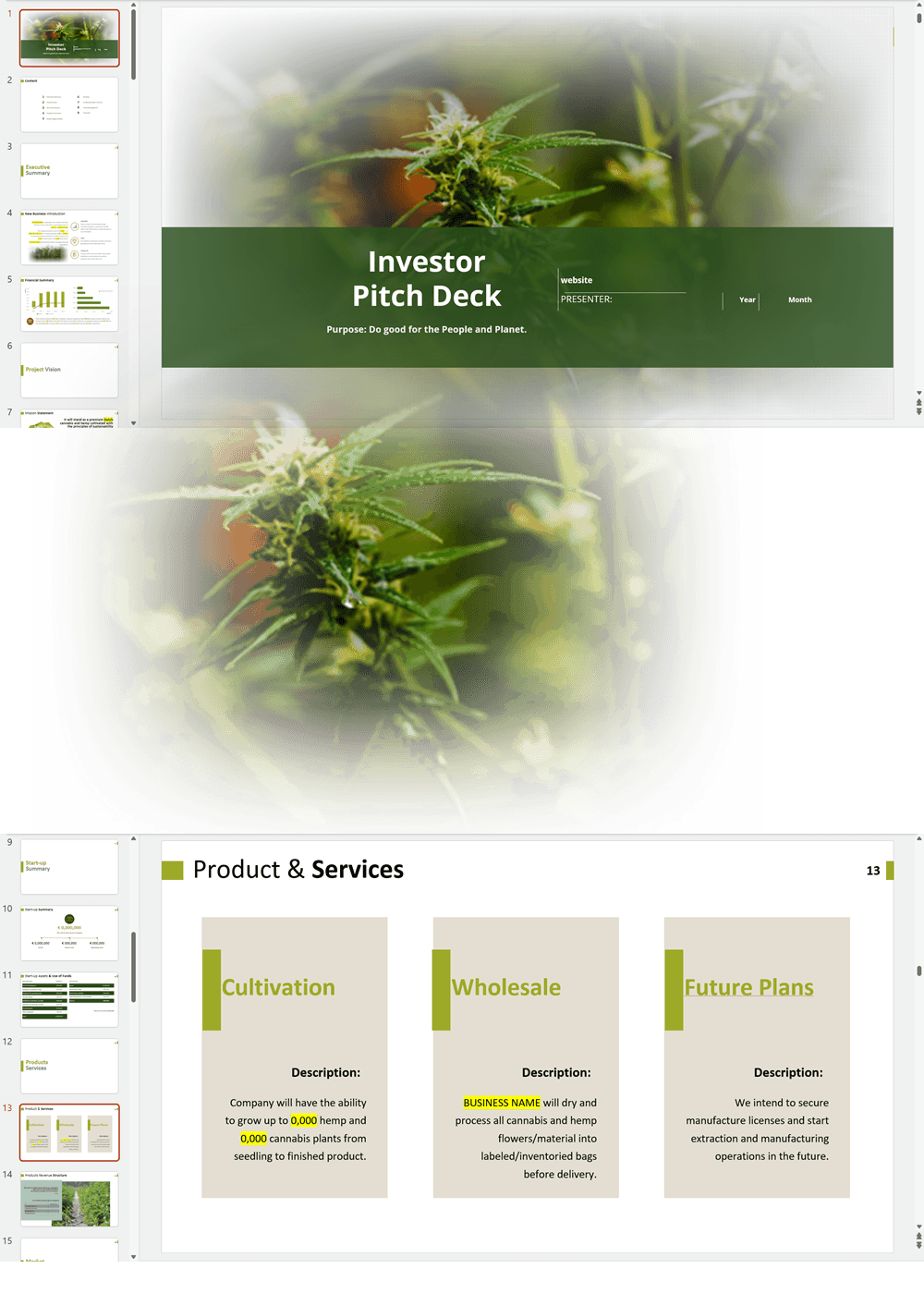
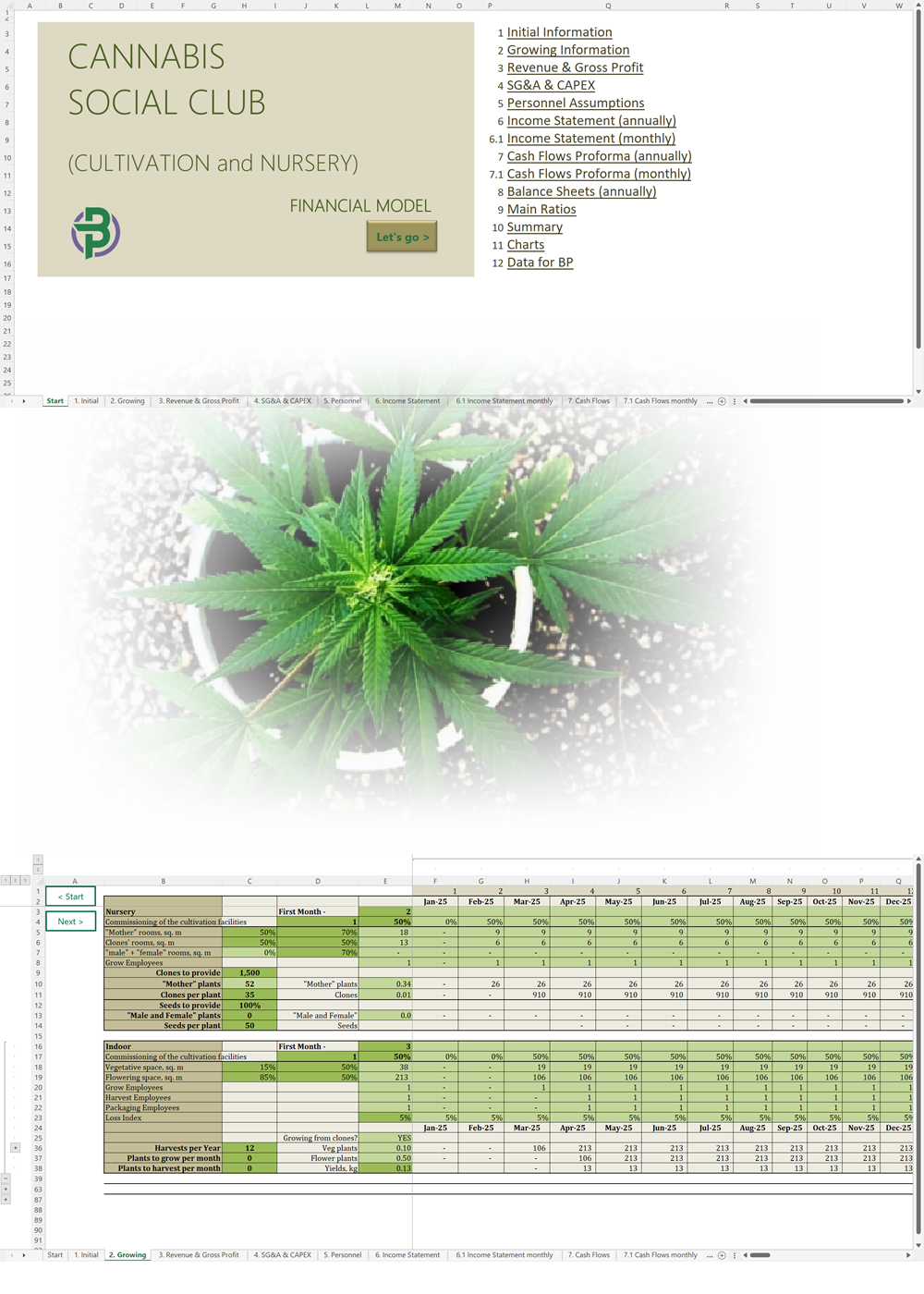
'70% ready to go' business plan templates
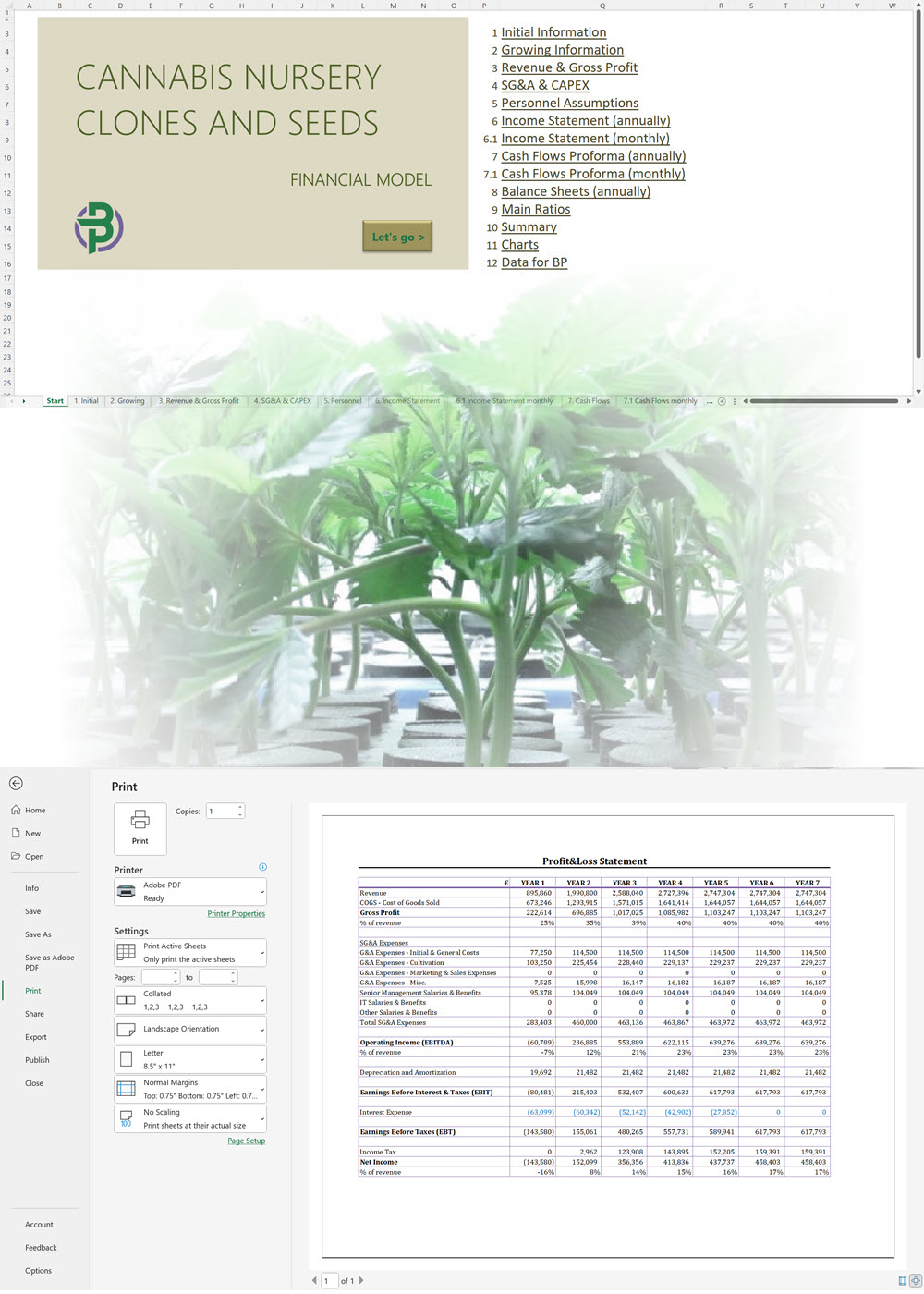
We offer a '70% ready-to-go' Cannabis Export Import Distribution Business Plan Template that will help you prepare a professional cannabis business plan document supported with expert financials and an investor pitch deck. All our templates are completely open and editable and you can use Word, Excel and PowerPoint to work with them.
It is essential that you follow Good Agriculture and Collection Practice (GACP) if you wish to export cannabis plant material to Europe. GACP covers all activities before the drying stage: cultivation, harvesting and collection of cannabis plant material. All raw material used in medicine in the EU is subject to GACP regulations – this is to ensure product safety and consistency for the consumer. The main principles of GACP are record keeping and consistency in how the plant is cultivated. For record keeping, you will need to have a Standard Operating Procedures (SOPs) document, outlining at minimum procedures for the following:
- quality assurance
- personnel and education
- buildings and facilities
- equipment
- documentation
- seeds and propagation material
- cultivation
- collection
- harvest
- primary processing
- storage and distribution
- Obtain the EU-GMP Certificate or a Written Confirmation.
The Good Manufacturing Practices (GMP) regulations covers drying and all the following manufacturing processes. An EU-GMP certificate is required to supply active pharmaceutical ingredients (APIs) – the component of a medication that produces the intended therapeutic effects – or finished medical cannabis products for the EU market. Alternatively, instead of obtaining an EU-GMP certificate, you may request a Written Confirmation from an EU competent authority which confirms that the GMP standards applied at your facility are equivalent to those applied in the EU. However, this requirement does not apply to certain countries and territories listed by the European Commission. Regarding the EU-GMP certificate, there are two options: partnering with existing EU-GMP-certified facilities or obtaining an EU-GMP certificate yourself. It should be noted that if you wish to simply export APIs or finished medical cannabis products, then it is not required that the starting material (the cannabis plant material) comes from an EU-GMP certified facility, though it does help. Requirements for obtaining an EU-GMP certificate include the following:
- Quality Management System (QSM) that documents all procedures and practices to ensure the quality of products.
- Standard Operating Procedures (SOPs) for all aspects of cannabis cultivation, harvesting, drying, trimming, processing, extraction, packaging, labeling, storage and destruction.
- Regular staff training on GMP guidelines, the QSMs, SOPs and other relevant topics.
- Facility must be clean, safe and suitable for cannabis product production.
- Equipment must be calibrated and maintained regularly to ensure accurate and consistent results.
- Documentation – all aspects of cannabis processing, including testing, must be documented.
- Regular testing to ensure products meet quality standards.
- Tracking system to track products from cultivation to distribution.
If you are planning to acquire the EU-GMP certification, you can follow these steps:
- Compare the existing practices at your facility with the EU-GMP requirements and identify which areas need to be improved.
- Write up SOPs for all processes. As a cannabis facility, this was likely already part of your cannabis license application.
- Using the SOPs, create training programs for all your staff so they are familiar with them.
- All manufacturing equipment must be qualified and validated.
- Carry out an internal audit to ensure compliance and address any issues that come up.
- Submit an official request for a GMP certification inspection to a competent authority of the particular European country (e.g. the BfArM in Germany).
- Prepare a Site Master File and identify the Qualified Person.
- Undergo the GMP inspection.
If the inspection is successful, the EU-GMP certificate will be issued. It is essential to maintain compliance with the GMP as period inspections will take place.
A marketing authorization allows to market a medicine in European Union Member States. To receive a marketing authorization, manufacturing sites must comply with EU-GMP which is why the previous step is important. A marketing authorization can be obtained via:
- Centralized procedure – A single application is submitted to the European Medicines Agency (EMA). This procedure may be the only pathway for obtaining a marketing authorization for certain medicines.
- Decentralized or mutual recognition procedure – A single application is made to one EU member state which will coordinate the application on behalf of the other member states. A decentralized procedure is applicable when the product has not been authorized in any EU country. A mutual recognition procedure is applicable when a product has already been authorized in at least one of the EU member state.
- National procedure – An application to the competent authority in an individual EU member state under its national procedure. This type of marketing authorization will only be valid in that EU member state.
- Carry out market research. Identify specific EU countries to which you wish to export cannabis. Research cannabis regulations in those countries.
There is no general single pathway for importing cannabis into Europe. Different countries have their own regulations for cannabis. You can find a wide range of articles on the cannabis markets in different EU countries on our website:
Identify your partners – an EU licensed manufacturing facility that will produce the finished product or the place where your product will be dispensed to patients (this could be a pharmacy in some countries) – and secure an agreement with them. Check which import and export permits are required, apply for and obtain them.
Cannabis Export Import Distribution Business Plan Sample (Australia)
We also offer a Cannabis Wholesaler Business Plan Template.
Should you have any questions, please do not hesitate to contact us.