Main steps in short:
- Understand regulatory and legal requirements. Do market research.
- Decide the type of cannabis business. Choose a location. Check local zoning regulations.
- Develop a solid business plan. Secure financing.
- Prepare the required documents. Pay application fee. Submit application.
- Keep track of your ongoing compliance requirements.
1. Understand the regulatory and legal requirements. Do market research.
Cannabis Legalization
On January 19th, 2017, Germany’s parliament (Bundestag) passed a law that officially made cannabis legal for medicinal purposes, allowing for severely ill German patients to receive prescriptions for medical cannabis treatment. This amendment became effective from March 2017, allowing people with qualifying conditions to legally obtain medical cannabis from pharmacies with a doctor’s prescription.
At first, the Federal Institute for Drugs and Medical Devices (BfArM) awarded a tender to cultivate cannabis domestically in Germany to three companies exclusively for distribution in German markets. On 22 March, 2024, the Cannabis Act (CanG) was approved by Germany’s upper house. It came into force on 1 April, 2024. It consists of the Consumer Cannabis Act (KCanG) – which legalized recreational cannabis and regulates the cultivation of cannabis at home and cannabis social clubs – and MedCanG which collects the regulations on medical cannabis into one law and established a purely authorization-based procedure instead of an award-based tendering process.
2. Decide the type of cannabis business. Choose a location. Check local zoning regulations.
3. Develop a solid cannabis business plan. Secure financing.
4. Prepare required documents and obtain the required licenses.
Cannabis Licenses Available in Germany
Anyone wishing to cultivate, produce, trade in, import, export, dispense, sell, obtain or acquire cannabis for medical purposes or for medical-scientific purposes, requires a permit from the BfArM. Cultivation permits are issued by the Cannabis Agency while permits for other activities are issued by the Federal Opium Agency.
The permit for establishing an adult-use cannabis cultivation association is issued by the competent authority chosen by each one of Germany’s federal states.
The following licenses are available along with the information and documents that must be submitted with the application:
A Medical Cannabis Cultivator license allows the cultivation of medical cannabis.
The following information must be submitted with the application:
- Surname, first name, and address of the applicant and all responsible persons and if applicable, the name and address of the company. For a legal representative of the applicant: the surname, first name, and address. For legal entities or unincorporated associations: the surname, first name, and address of the person authorized to represent the applicant by law, statute or partnership agreement.
- A copy of the current and complete extract from the commercial register/association register.
- Proof of application for a certificate of good conduct to be presented to an authority in accordance with Section 30, Paragraph 5 of the Federal Central Register Act.
- Proof of the required expertise for each responsible person, demonstrated pursuant to Section 15, paragraph 1 of the Medicinal Products Act.
- A description of the location of the business premises, by town, if possible with the field name, as well as the street, house number, building and part of the building.
- The type of cannabis intended to be used for the permitted activities.
- The complete “Declaration form for medical cannabis managers at companies” for each responsible person.
- Description of the suitable measures and security precautions to prevent unauthorized access to medical cannabis.
- Room or site plans of the cultivation areas.
- Information on the planned destruction of cannabis flowers and plant parts that rule out recovery.
- Description of the cultivation and production process.
- Information on whether germ reduction is part of the production and if so, which germ reduction process shall be used.
- Confirmation of compliance with GACP (Good Agricultural and Collection Practice).
- Valid manufacturing permit/GMP (Good Manufacturing Practice) certificate from the responsible state authority for the production associated with cultivation of medical cannabis flower (e.g. drying or other steps if necessary).
- Information on propagation and cultivation.
- Information on the composition of the total cultivation area (e.g. planned number of plants/cultivation cycles/rooms).
A Medical Cannabis Dealer license allows to participate in medical cannabis trade.
The following information must be submitted with the application:
- Address and name of the company as well as the name and address of all persons authorized to represent the company by law, statute or partnership agreement.
- Copy of the current and complete extract from the commercial register.
- Address of the business premises participating in the medicinal cannabis trade, if applicable, including details of the building and part of the building as well as details of the contact persons with telephone number, email address and fax number if applicable.
- For the person responsible for the medicinal cannabis trade:
- Complete declaration form.
- Proof of expertise according to Section 7 Paragraph 3 MedCanG.
- Proof of application for a certificate of good conduct to be presented to an authority in accordance with Section 30, Paragraph 5 of the Federal Central Register Act.
- If carrying out pharmaceutical wholesale, a copy of the wholesale license in accordance with Section 52a AMG.
- List of the respective types of cannabis for medical purposes or cannabis for medical-scientific purposes that are to be handled.
- Indication of the type of trade – purchase or sale; import or export; in case of commercial activity, whether it is domestic or foreign trade.
- In case of use for scientific purposes, a detailed explanation of the purpose pursued.
A Medical Cannabis Manufacturer license allows the extraction, manufacture, preparation, treatment or processing, purification and transformation of medical cannabis.
The following information must be submitted with the application:
- Information about the applicant/company:
- Name and domestic business address of the applicant/company.
- Description of all affected business premises according to their town, street, house number, building and part of the building.
- Details of contact persons with telephone number and email address, fax number if applicable.
- Copy of the current and complete register extract.
- Surname, first name and address of the applicant.
- Certificate of good conduct for submission to an authority in accordance with Section 30, Paragraph 5 of the Federal Central Register Act for all authorized representatives.
- For each responsible person:
- Complete “Declaration form for medical cannabis managers at companies”.
- Certificate of good conduct for submission to an authority in accordance with Section 30, Paragraph 5 of the Federal Central Register Act.
- Proof of expertise according to Section 7 Paragraph 3 MedCanG.
- List of types of medical cannabis:
- The type of medical cannabis or substance or its sale using the name listed in the MedCanG.
- For preparations, their full name, authorization number as well as the substances contained and their contents.
- For foreign preparations, a copy of the outer packaging.
- Indication of the type of trade: purchase or sale; import or export; in case of commercial activity, whether it is domestic or foreign trade.
- For scientific purposes, a detailed explanation of the purpose pursued.
- For manufacturing substances:
- Description of the manufacturing process.
- Details of all starting materials by type and quantity.
- Details of the intermediate and end products.
- Details of the theoretical yield.
- Details of the individual manufacturing steps.
- For manufacturing preparations:
- Details of all starting materials by type and quantity.
- Details of the individual manufacturing steps.
- Details of the theoretical yield.
- Labelling of all intermediate and final products used and/or manufactured.
- Details of further purpose – whether preparations are for domestic use or for export.
- If for export, whether the manufacturer or another company will carry out the export, and the name and registered office of the marketing authorization holder responsible for the importing country.
An Adult-Use Cannabis Cultivation Association is a registered non-profit association or cooperative that has the exclusive purpose of communal non-commercial cultivation and distribution of cannabis for recreational use by members of the association.
The following rules apply to cannabis cultivation associations:
- Members must be at least 18 years old and must have a residence or habitual abode in Germany.
- An association can have a maximum number of 500 members.
- An individual can be a member of only one cultivation association.
- Samples of the cannabis cultivated and the propagating material must be regularly taken and examined.
The following information must be submitted with the application:
- Name, telephone number and electronic contact details as well as the address of the registered office of the cultivation association.
- Competent registry court and the registration number of the cultivation association.
- First name, surname, date of birth, address and electronic contact details of the board members and all authorized representatives of the cultivation association.
- First name, surname, date of birth, address and electronic contact details of all paid employees of the cultivation association who have access to cannabis and propagation material.
- A certificate of good conduct issued no more than three months before the application for permission to be submitted to an authority in accordance with Section 30, Paragraph 5 of the Federal Central Register Act, as well as information from the Central Trade Register in accordance with Section 150 (1) Sentence 1 of the Trade Regulation Act, issued no more than three months before the application for permission for each member of the board and for each other person authorized to represent the cultivation association.
- Estimated future number of members of the growers’ association.
- Location of the protected property of the cultivation association by town, street and house number, if applicable, indication of the field name, the name of the building and the part of the building.
- Size of the cultivation areas and greenhouses of the cultivation association in hectares or square metres.
- Quantities of cannabis in grams, separated into marijuana and hashish, expected to be cultivated and distributed annually.
- Description of security and protective measures taken in accordance with Section 22 Paragraph 1 of KCanG.
- First name, surname, date of birth, address and electronic contact details of the prevention officer appointed pursuant to Section 23, Paragraph 4, Sentence 2 of KCanG as well as evidence of counselling and prevention knowledge as required pursuant to Section 23, Paragraph 4, Sentence 5 of KCanG.
- Health and youth protection concept to be drawn up in accordance with Section 23 Paragraph 6 of KCanG.
The permit is valid for seven years and can be extended after a period of at least five years.
In addition to these three licenses, there are also licenses available for:
- Cultivation of cannabis for medical and scientific purposes.
- Clinical trials.
- University institutions.
- Non-university scientific institutions.
Pharmacies can deliver medical cannabis to patients.
For import or export of cannabis, an additional permit is required from the BfArM for each import or export. Details on how to apply for these permits and what information must be provided with the application, can be found on the official website of BfArM.
5. Keep track of your ongoing compliance requirements.
Medical cannabis licensees are required to maintain records, separate for each business premises and for each type of cannabis for medicinal or medical-scientific purposes. The records must be kept for three years.
Additionally, medical cannabis licensees must report electronically to the BfArM by January 31 of each calendar year, separately for each business premises, all stocks and manufacturing processes.
Cannabis Taxes in Germany
There is no specific excise tax on cannabis in Germany.
Cannabis Advertising in Germany
In Germany, medical cannabis may be advertised to specific healthcare professionals. There is a general ban on advertising or sponsoring of adult-use cannabis and cultivation associations.
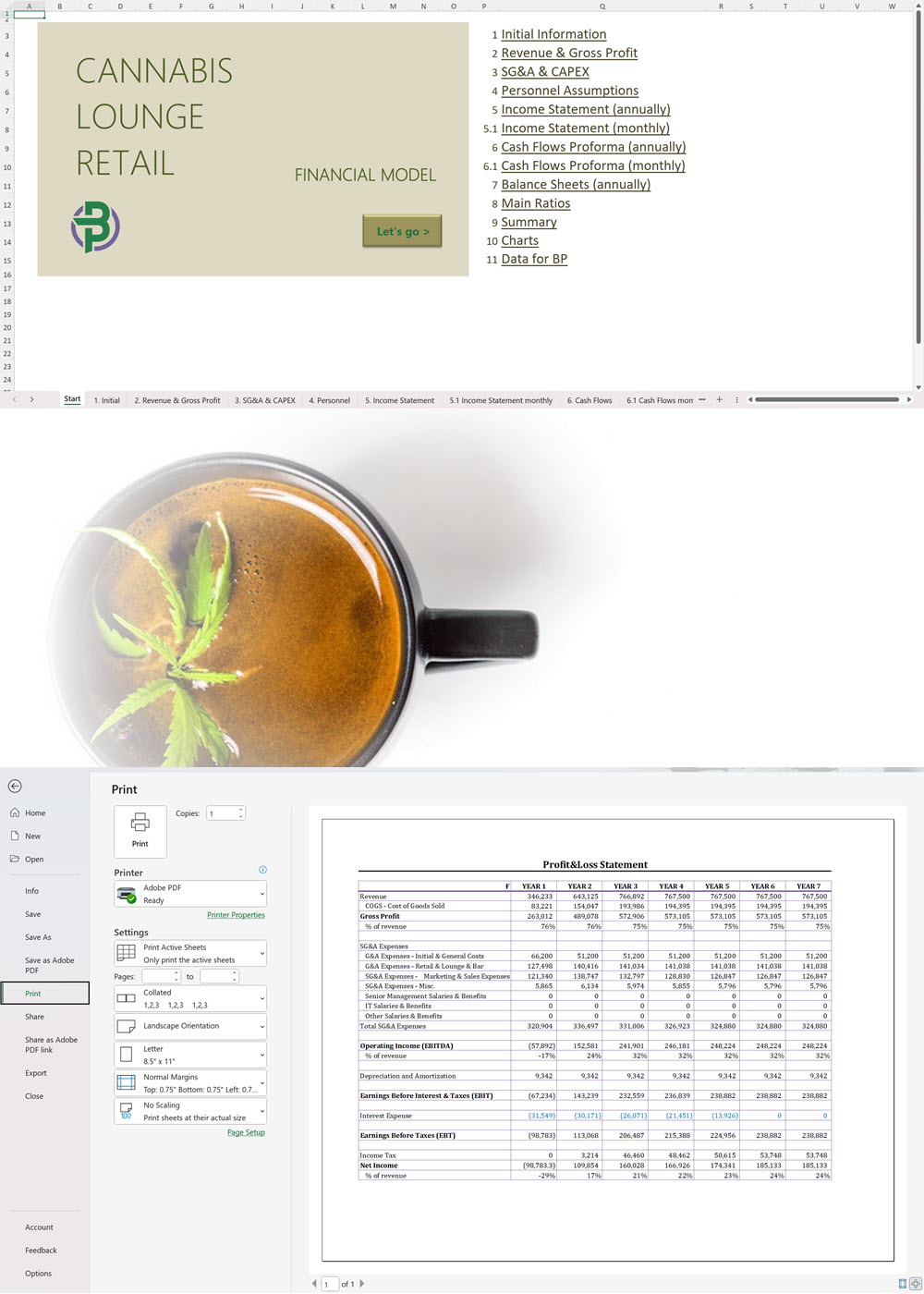
Cannabis Cultivation Business Plan Sample, Germany
'70% ready to go' business plan templates
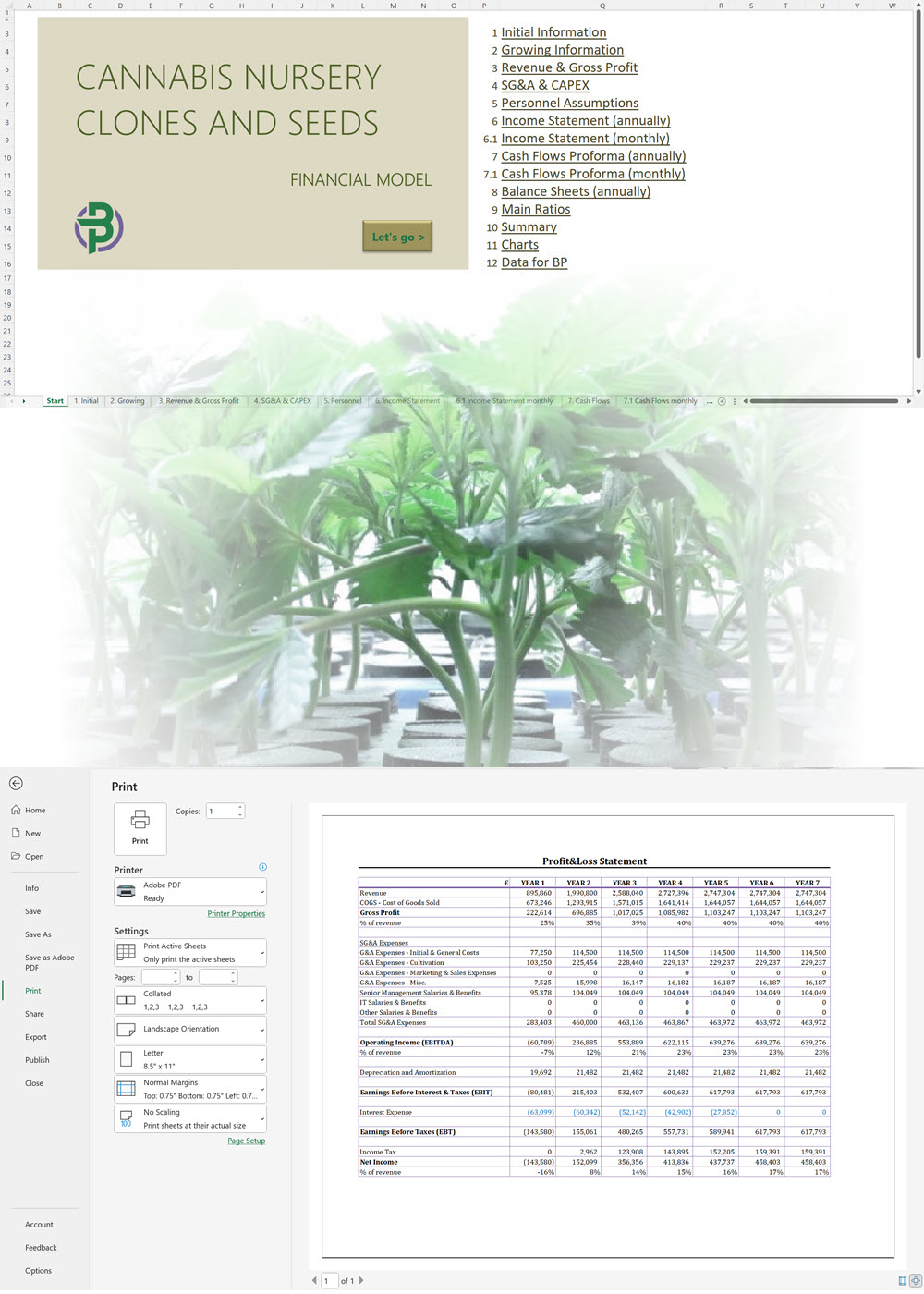
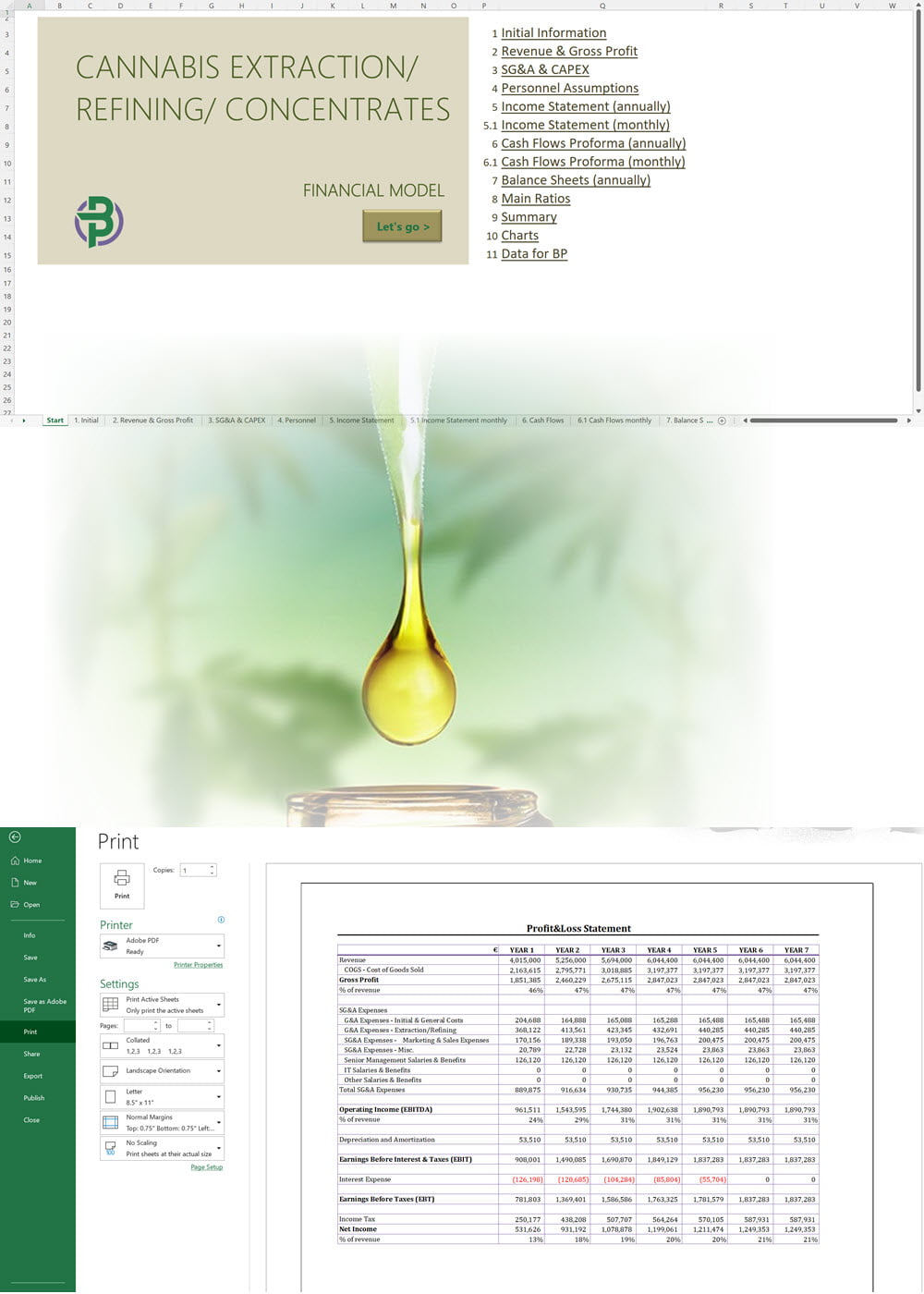
Our cannabis financial models and cannabis business plan templates will help you estimate how much it costs to start and operate your own cannabis business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates.
Best Selling Templates
-

Cannabis Cultivation Business Plan Template
Price range: €75 through €350 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Dispensary Investor Pitch Deck Template
€75 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Financial Model All in One
€250 Select options This product has multiple variants. The options may be chosen on the product page
Hemp/CBD business plan templates are also available at hempcbdbusinessplans.com.
Templates for other countries are available at cannabusinessplans.com.