Hemp Legalization in the UK
Most cannabis-derived products are controlled substances under the Misuse of Drugs Act, but some types of CBD oil are legal to sell as health supplements. CBD oils approved for sale in the UK must not contain more than 0.2% of THC, the chemical responsible for the ‘high’ effect.
The legislative controls identified above apply to cannabis plants cultivated for the production of drug material (e.g. hemp fibre or oil). Cultivation or possession of cannabis plants cannot lawfully be undertaken without the requisite Home Office Licence.
Licences are granted to enable the use of non-controlled parts of the plant (i.e. seeds and fiber/ mature stalk only) and the policy does not allow for use of ‘green’ material – i.e. the leaves and flowers as these are controlled parts of the plant. The Home Office only issues licenses for cultivation of plants from approved seed types with a THC content not exceeding 0.2%.
The Misuse of Drugs Act 1971 treats hemp flower as a “controlled substance, regardless of the THC content” and it is therefore illegal. In June 2023, the Court of Appeal stated that a hemp flower with less than 0.2% THC is not a narcotic drug and should not be treated in the same way as high-THC cannabis products. This ruling may lead to a legal, low-THC flower market in the UK in the future.
Products containing CBD oil or extract are classified as novel foods and must obtain a novel food authorisation from the Food Standards Agency (FSA) before they can be placed on the market. Hemp seeds, hemp seed oil, defatted seeds, and hemp seed flour are not classified as novel foods.
Any single bottle or packet of CBD must not contain more than 1mg of THC, regardless of its size. In October 2023, the FSA published advice on CBD which recommended adults to “limit their consumption of CBD from food to 10mg per day” – a fall from the previous limit of 70 mg per day set in 2020. The British Retail Consortium (BRC) stated that its members would “follow any and all FSA advice on the sale of products containing CBD”.
Industrial Hemp Licensing in the UK
An ‘industrial hemp license’ from the Home Office is required to cultivate low-THC cannabis plants (with THC content no more than 0.2%) for the production of hemp fiber for industrial purposes or the obtaining of seeds for oil. This license will allow the license holder to only use non-controlled parts of the plants (“seeds and fiber/mature stalk only”) and it does not allow the use of ‘green’ material (leaves and flowers). The holder of the license can sow only EU/UK approved seed varieties with less than 0.2% THC content.
- Read the industrial hemp application guidance published by the Home Office.
- Register with the Home Office Controlled Drugs Licensing System.
- Ensure that every person named on the application form “holds a valid enhanced Disclosure and Barring Service (DBS) check” which must be performed by Security Watchdog. A DBS from any other company will not be accepted. The DBS check must not be older than 3 years.
- Submit your annual hemp return.
Important dates such as when the application for an industrial hemp license opens and the latest date by which you should apply as well as other details can be found on the Home Office website, updated for the relevant growing season.
Details of information that has to be included in the application can be found here. The summary is:
- Details of the people to be included on a license
- Farm business/company or organization details
- Licenses requested
- Details of an enhanced DBS check being obtained within the last three years from Security Watchdog for drug licensing purposes
- Premises details including any physical security arrangements and details of cultivation locations
- Record keeping and audit
- Supplier/customer details
- Documents such as aerial photos and site maps, photos of record keepings, copy of seed invoices etc.
- Fee payment and refund details
- Where the license has to be sent to
New license application fee: £580
License renewal fee: £326
- Licenses for industrial hemp are “valid for 3 growing seasons”. Licenses are always issued to expire on 31 December.
- Once the industrial hemp cultivation license is approved, an email will be received with instructions for payment. The license will only be issued after payment.
- An annual hemp grower statement must be submitted by 1 March every year, regardless of whether cultivation took place in that year.
- The Home Office should be informed of any changes made to planting locations.
If the application is refused, the applicant can submit “any further representations in respect of the decision” within 30 days of the date of the refusal letter.
Changes introduced from 2025 growing season:
The National Farmers’ Union (NFU) secured changes to hemp licensing rules. License holders will be able to grow hemp “anywhere on a licensed farm” and the maximum period for a license will increase from three years to six years. Growers will also be able to apply for a license with a deferred start date of up to a year.
Hemp-derived CBD Products in the UK
CBD Novel Foods
In the UK, CBD can often be found in products such as oils, drops, tinctures, sprays, gel capsules, sweets, bread and other bakery products, drinks, and other food and food supplement products.
The FSA allowed CBD products to be sold in England, Wales and Northern Ireland until 31 March 2021 as long as:
- “The food was safe.
- The food was not contaminated with other cannabinoids.
- The food was correctly labelled.
- The food was placed on the market in England, Wales or Northern Ireland before 13 February 2020.”
In January 2019, CBD extracts and food products containing CBD extracts were classified as ‘novel foods’ and from 13 February, 2020, no new CBD products were allowed to be put on the market without the necessary authorisation.
By 31st March 2021, all UK CBD companies selling products for consumption had to submit a Novel Food application to the UK’s FSA. After this deadline, any CBD products on the UK market without an FSA ‘validated’ novel food application had to be withdrawn. CBD products which have an FSA ‘validated’ novel food application were added to the ‘Public List’ and allowed to remain on sale in Great Britain while the authorisation process is still ongoing. ‘Validated’ means that their sale is tolerated but “they are not ‘fully authorized’ for sale”. After being validated, the applications move on the risk assessment and risk management stages, before they can be granted an authorisation.
Guidance on the application requirements is set out here as well as in this document published by the EFSA (European Food Safety Authority) which is still relevant as UK’s approach is based on EU processes.
Following safety assessments, on July 3, 2025, the FSA recommended CBD food business operators to reduce the concentration of CBD in products so that it is not greater than the acceptable daily intake (ADI) of 10mg/day CBD for pure form ≥ 98% CBD and the safe upper limit of 70 µg/day Δ9-THC in foods. Though products that are not reformulated based on these new limits can continue to await the outcome of the novel food application, the FSA suggests that CBD companies currently in the process of obtaining an authorisation for their products may reformulate their products to make the authorisation process more efficient.
In 2024, safety assessments of three novel applications were published and approved by the FSA. Since the start of 2025, another five new safety assessments for CBD novel food applications have been published and approved by the FSA. All of these five products adhere to the limit of 10mg/day of CBD. These applications can now move on to the risk management stage.
CBD Cosmetic Products
Cosmetic products that include CBD must follow the Regulation 2009/1223 (UK Cosmetics Regulation). According to a position paper from the Cosmetic Toiletry and Perfumery Association (CTPA) published on July 2022, seeds and any extracts or oil from the seeds “can be used in finished cosmetic products” as long as their use and the finished product are safe. Cannabis leaf extracts and plant-derived CBD can also be used in cosmetic products if they meet the requirements which are specified in the position paper. The finished cosmetic product should not contain more than 1mg of controlled substances per container, “irrespective of size and not based on a specific concentration”.
CBD Medicinal Products
On April 14, 2025, the Medicines & Healthcare Products Regulatory Agency (MHRA) confirmed that CBD products that make a medicinal claim are considered to fall under the definition of a medical product in The Human Medicines Regulations 2012. Such products must hold a Marketing Authorisation in order to be sold, supplied or advertised in the UK.
CBD Café, Restaurant and Retail
No specific license is required if the retailer sells only hemp-derived CBD products. Retailers have to ensure however, that they sell CBD products from suppliers who are authorized by the FSA under the Novel Food Application scheme. Any product being sold must not contain more than 1mg of THC per package.
Importing CBD
According to the law firm Kingsley Napley, “there are no strict requirements for importing and selling CBD products in the UK” if the content of THC in them is below the detection limit. This, however, does not apply to CBD flowers.
As CBD food products and food supplements are classified as novel foods, an authorization from the FSA must be obtained before imported CBD food products can be sold on the UK market. CBD foods where “CBD is derived from cold pressed hemp seed oil or flour” are excluded from novel food regulations and therefore no authorization is required to sell them.
When importing CBD products, all laws and regulations regarding import into the UK must be followed such as submitting an import declaration and obtaining a EORI (Economic Operators Registration and Identification) number. Guidance on preparation for importing CBD products into the UK can be found here.
According Savills, CBD oils are imported into the UK primarily from the US and Eastern Europe.
UK Hemp CBD Market
The agriculture industry in the UK has started to recognise the increasing value of hemp as a crop and is expected to increase output and processing capabilities by 200% in the next 5 years. In 2021, the first stage of a project named “HEMP-30” gained funding support from the UK government. It aims to boost the amount of land on which cultivated industrial hemp is grown in the UK to 80,000 hectares by 2031. This is expected to add £700 million to the UK economy and sequester or displace 1 million tonnes of carbon dioxide annually.
In 2022, hemp was grown on an area of approximately 800 hectares with an estimated 30% of it cultivated for oilseed. It is estimated by the HEMP-30 project that in 2022, 38% of the hemp produced went into construction and nearly 47% of hemp biomass was used on farms for primarily animal bedding
The number of hemp licenses in the UK has increased from just six in 2013 to 139 in 2023.
In hemp varieties grown for seed or fiber use, the plants are grown very closely together and a very dense biomass product is obtained, rich in oil from the seeds and fiber from the stalks and low in THC content.
Much of the hemp grown in the UK in recent decades has been used in the construction industry in various forms, mainly as non-structural building materials and wall insulation. Hemp’s natural insulating properties and amazing durability make it a viable alternative to traditional materials in terms of technical quality.
The Cannabis Trades Association UK estimated that UK CBD consumption doubled from 2016 to 2017, rising from 125,000 to 250,000 users. Although CBD, making medicinal claims, needs to receive a marketing authorisation from the MHRA, many companies have set up CBD companies categorised as food supplements.
In that time, the market value for CBD oil in the UK has gone from almost nothing to €57m per annum. The health and well-being market looks likely to capitalize on growing public interest, with CBD-based snacks, suppliments and even skincare products making their way onto shelves.
A YouGov survey found that nearly one-in-ten adults in the UK have tried CBD products. The most popular CBD product form is oil – 58% of CBD users among survey respondents – which is followed by vape products (21%) and creams (11%). Around 61% of CBD consumers use it for medical purposes, of which 71% use it to treat pain while 38% use it to treat anxiety and depression.
There are currently no authorised CBD extracts or isolates on the UK market but as of July 2025, 8,147 novel food applications for CBD products have been validated – this means that the application contains all necessary information to proceed to the risk assessment and risk management stages, and then onto authorisation.
In 2021, UK’s CBD market was valued at £690 million which is an increase of £314 million since 2019 according to a report by the Association for the Cannabinoid Industry, making it the largest consumer cannabinoid market in the world after US. It is predicted that by 2025, the CBD market in the UK will be worth around £1 billion.
Source: hempcbdbusinessplans.com
How Can HempCBDbusinessplans Templates Help Start or Grow Your Hemp CBD Business?
A business plan is essential for securing funding. It will also help you understand how much money it will take to start a hemp CBD business and how much profit it could make. A well-structured business plan can make all the difference in demonstrating your proposed hemp CBD business’ compliance with regulations and its operational viability.
Our complete hemp CBD business plan template package is everything you need to create a professional business plan for a hemp CBD business with expert financials and projections. A complete hemp CBD business plan template package includes:
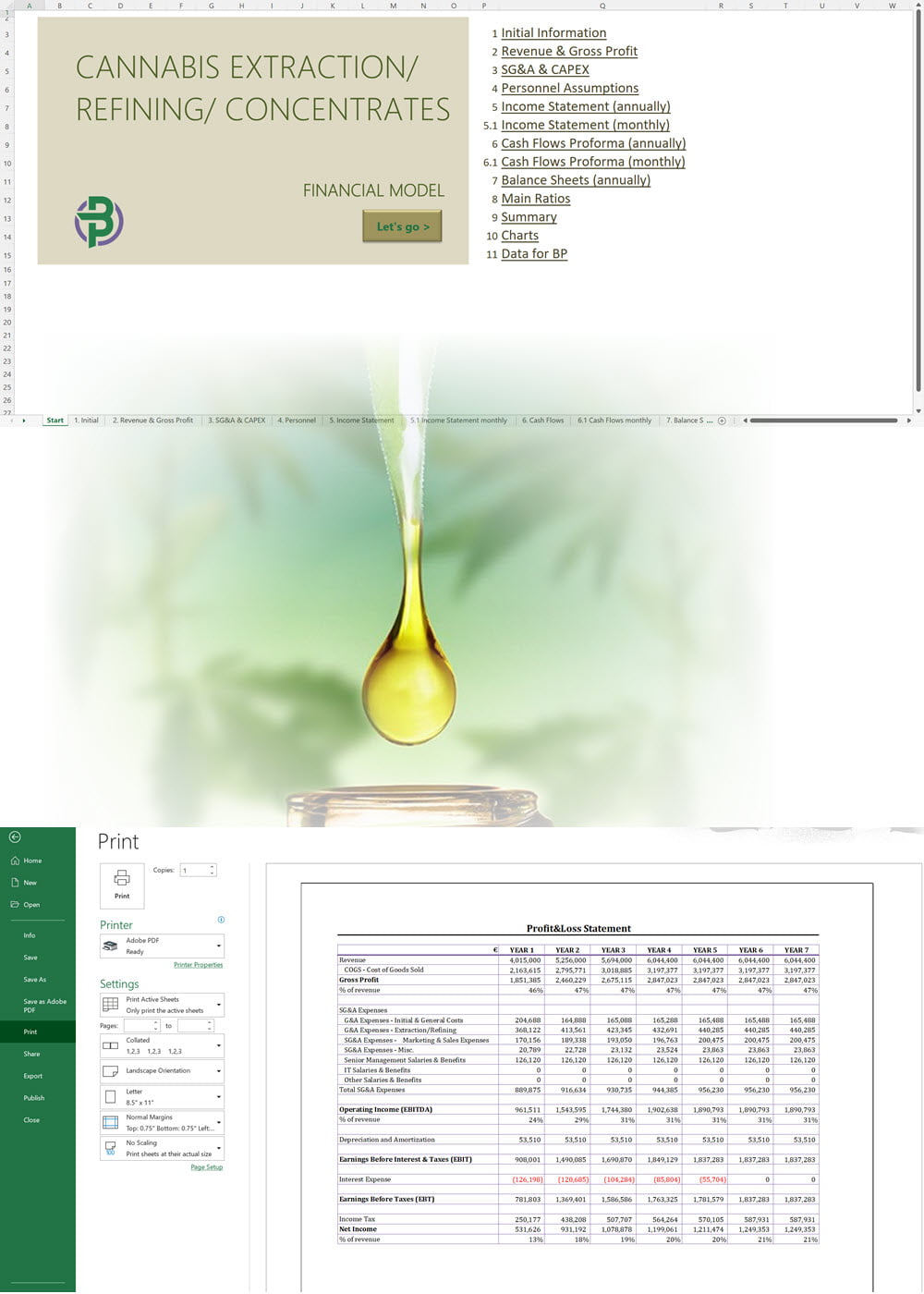
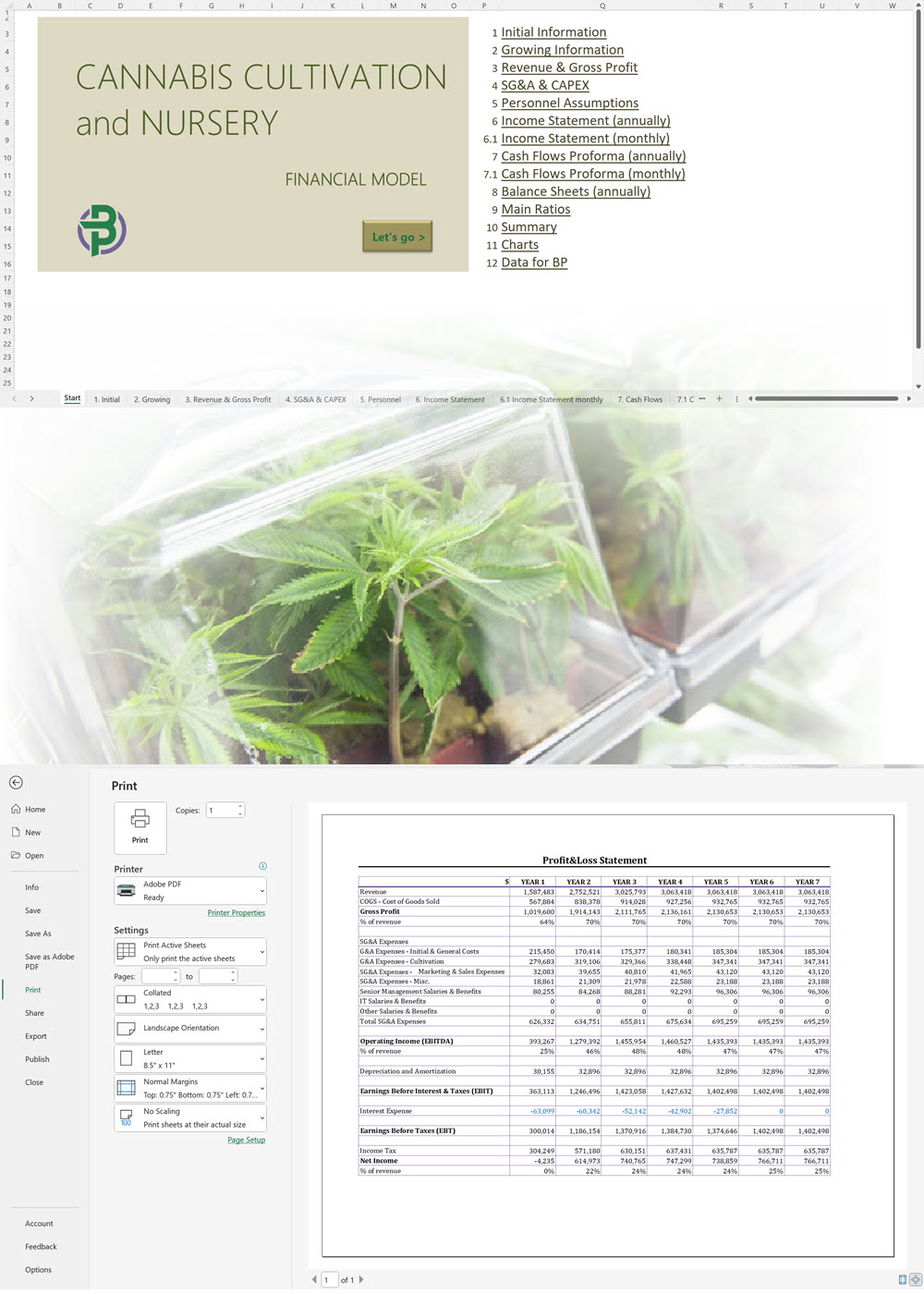
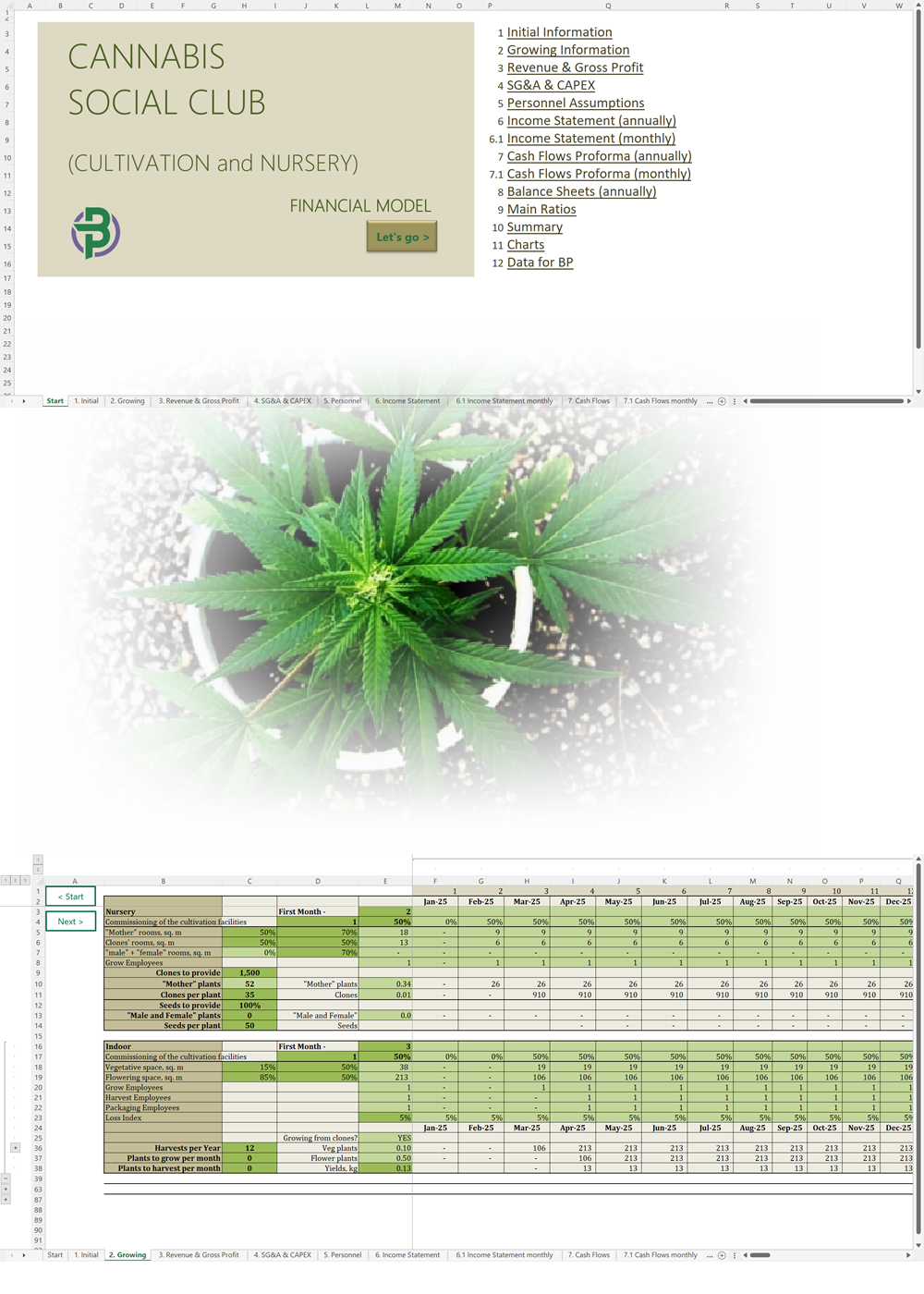
- Excel Financial Model: change variables and immediately see the impact, break down operational and capital costs, know how much it will take to get into the business and the potential profits.
- Word Business Plan: value proposition, market analysis, marketing strategy, operating plan, organizational structure, financial plan and more.
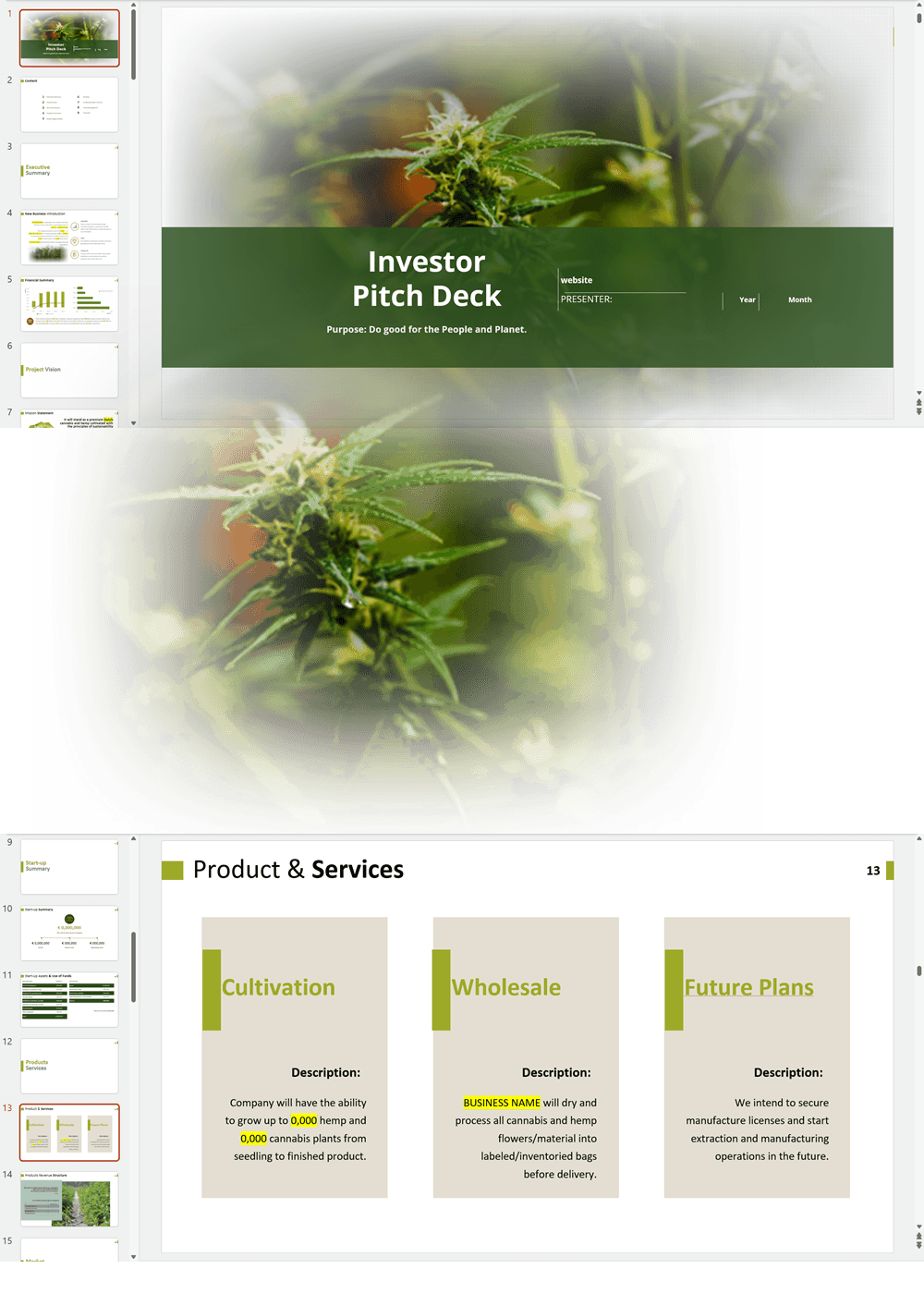
- PowerPoint Pitch Deck: provide a quick overview of your hemp CBD business plan.
'70% ready to go' business plan templates
Our hemp/CBD financial models and business plan templates will help you estimate how much it costs to start and operate your own hemp/CBD business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates.
Hemp/CBD Products Retail and Lounge Pitch Deck Template Sample, the UK
Best Selling Hemp Templates
-

Hemp Cultivation + CBD Oil Extraction and/or Fiber Products Financial Model
€150 Select options This product has multiple variants. The options may be chosen on the product page -

CBD Retail Investor Pitch Deck Template
€70 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis and Hemp Vertically Integrated Business Plan Template
Price range: €100 through €550 Select options This product has multiple variants. The options may be chosen on the product page
For more information on the hemp industry and hemp/CBD business plan templates, visit hempcbdbusinessplans.com.
Cannabis business plan templates are available at cannabusinessplans.com.