Medical Cannabis Legalization in the UK
A licence from the Home Office is required to produce, possess, supply, cultivate, import or export cannabis and cannabis-based products for medicinal use in humans (CBPM).
On July 26, 2018, the then Home Secretary announced that has had decided to move cannabis-based medicinal products to schedule 2 of the Misuse of Drug Regulations 2001 which would make it legal to prescribe cannabis-based medicines. On November 1, 2018, the medicinal cannabis was re-assigned to Schedule 2 and the new regulations allow specialist clinicians to legally prescribe cannabis-derived medicinal products to patients.
Cannabis is a Class B controlled drug under Part II, Schedule 2, of the Misuse of Drugs Act 1971 (MDA 1971). It is also listed in Schedule to the Misuse of Drugs Regulations 2001 (MDR 2001) and designated under the Misuse of Drugs (Designation) (England, Wales and Scotland) Order 2015 (2015 Order). As such, it is unlawful to possess, supply, produce, import or export this drug except under a Home Office licence. It is also an offence to cultivate any plant of the genus Cannabis except under a Home Office licence.
Each grower must be individually licensed.
Cannabis-based products for medicinal use in humans (CBPMs, also known as ‘specials’) are defined as “a preparation or other product, other than one to which paragraph 5 of part 1 of Schedule 4 applies which:
(a) is or contains cannabis, cannabis resin, cannabinol or a cannabinol derivative (not being dronabinol or its stereoisomers);
(b) is produced for medicinal use in humans; and
(c) is a medicinal product, or a substance or preparation for use as an ingredient of, or in the production of an ingredient of, a medicinal product.”
How to Start a Cannabis Business in the UK
No licence is required for CBPMs to be sold by registered pharmacies or retail pharmacy businesses as they “are able to produce/supply in accordance with their practice” as stated in the guidance from the Medicines and Healthcare products Regulatory Agency (MHRA).
A Controlled Drugs Licence is required for the cultivation, production, supply and possession of cannabis. It is issued by the Home Office. Home Office has an application guide for the Controlled Drugs Licence.
Fees:
Possess controlled drugs: £3,133
Supply, or offer to supply, controlled drugs: £3,655
Produce preparations containing controlled drugs: £4,178
Cultivate cannabis: £4,700
A Manufacturer’s (Specials) Licence allows to manufacture unlicensed medicines such as CBPMs as well as import unlicensed medicinal products from outside the EEA. It is issued by MHRA. MHRA has guidance for applicants and holders of a Manufacturer’s Licence.
Application fee: £6,019. More information about fees can be found on MHRA’s website.
A Wholesale Dealer’s Licence is required to sell or supply CBPMs to “anyone other than the patient using the medicine”. It also allows to import unlicensed medicinal products from EEA member states. The applicant must comply with good distribution practice (GDP) and pass regular GDP inspections of their site to obtain a Wholesale Dealer’s Licence. It is issued by MHRA. MHRA has guidance for applicants and holders of a Wholesale Dealer’s Licence.
Application fee: £1,983. More information about fees can be found on MHRA’s website.
Additionally, to export or import CBPMs, a single consignment export or import licence is required. An export or import licence needs to be obtained for each individual shipment. It is issued by the Home Office. Home Office has an application guide for Export and Import Licences.
Single consignment export licence: £24
Single consignment import licence: £24
From March 2nd, 2020, new regulations were introduced which allow wholesalers to import Specials in larger volumes and hold supplies for future use so patients with prescriptions can receive their medicine faster.
Prior to selling a medicine or prescribing it in the UK, it must receive a marketing authorisation from the MHRA.
Regulatory Framework & Access to Medical Cannabis
Typically, before a medicine can be sold or prescribed in the UK, it must receive a marketing authorisation from the MHRA. The marketing authorisation is issued only if clinical trials of a medicine have demonstrated that it successful treats the condition it was developed for, has acceptable side effects, and meets quality and safety standards. As of January 2026, only two cannabis-based medicines – Sativex, Epidyolex, and Nabilone – have a UK marketing authorisation.
However, under certain circumstances healthcare professionals can prescribe medicines that do not have a marketing authorisation to an individual patient. It is through this pathway that many cannabis-based medicines are currently being prescribed to patients in the UK. The majority of private clinics offering cannabis prescriptions specialize in medical cannabis and carry out consultations online, via video calls, with the product then delivered to the patient by post.
A range of preparations containing delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD) with differing percentages of the active constituents. The product is to be specified by brand/supplier; cannabis strain and content of THC/CBD (and ratio of THC/CBD where relevant), as appropriate.
All cannabis-based products except from Sativex, Epidyolex, and Nabilone are unlicensed medicines.
Available on a named patient basis when there is “clear published evidence of benefit or UK Guidelines and in patients where there is a clinical need” which cannot be met by a licensed medicine and where established treatment options have proven to be ineffective.
Medical cannabis can only be prescribed by doctors listed on the ‘Specialist Register’ of the General Medical Council.
Medical cannabis products must meet the requirements of the MHRA specials guidance and the additional cannabis guidance. The General Medical Council (GMC) has also published guidance on prescribing unlicensed medicines.
NHS guidance states that since the majority of cannabis-based medicines do not have a marketing authorisation, they are unlikely to be considered as a first line treatment and prescribers should always consider prescribing medicines that are licensed for the patient’s specific condition.
CBPMs can be purchased in the UK by:
- Doctors on the GMC Specialist Register
- Registered pharmacies or retail pharmacy businesses
- Specialist Importer with a Home Office import and Controlled Drugs domestic license as well as an MHRA license
- Licensed wholesale dealers for supply to any of the above
As CBPMs are unlicensed medicines, if they are imported or manufactured in the UK, they can only be sourced from suppliers and manufacturers licensed by the MHRA or an equivalent organization in the country from where the product is imported.
Medical Cannabis in the UK: Market Growth, Patient Access, and Supply
The UK medical cannabis market has experienced significant growth since the legalization of CBPMs. in 2018. Valued at an estimated £233 million, it is now the second-largest medical cannabis market in Europe – second only to Germany – and shows strong potential for continued expansion both domestically and internationally.
Patient Statistics
Although initial adoption was slow, patient numbers have accelerated in recent years. In the beginning of 2023, it was estimated that the drug had only been prescribed to 25,000 patients since it was legalized for medical use. However, by February 2024, private clinics such as Releaf reported enrolling approximately 3,000 new patients per month. It was forecasted that 62,690 patients will be using medical cannabis in the UK by the end of 2024, generating £205 million in sales. In 2025, an estimated 80,000 patients were using medical cannabis. By 2028, this number is projected to increase to 141,000 patients with the market value estimated at around £500 million.
The Department of Health and Social Care estimates that as many as 3 million patients with chronic pain could be eligible for cannabinoid treatments across the UK, indicating substantial unmet demand.
Prescription Trends: NHS and Private Sector
Prescribing activity has risen steadily across both public and private channels.
The number of licensed cannabis-based medicines prescribed to patients through the NHS doubled since 2018, reaching 5,192 units in 2023 according to data from NHS Business Service Authority. From 2018 to September 2024, the NHS issued a total of 24,395 prescriptions for licensed cannabis-based medicines. In the 12 months from February 2024 to January 2025 alone, the NHS issued 5,413 prescriptions for licensed cannabis-based medicines.
The private sector has driven the majority of prescribing volume as unlicensed CBPMs can be obtained only through private prescriptions. According to data from NHS Business Service Authority, between November 2018 and July 2022, 89,239 private prescriptions for CBPMs were issued. Inside that time period, a dramatic surge occurred between 2020 and 2021 when prescriptions increased almost tenfold from 4,469 to 42,393. Growth continued through the first half of 2022 with around 40,000 prescriptions issued – equal to the total volume of prescriptions issued for the entire of 2021. Meanwhile, data from the CQC (an independent regulator of health and social care in England) shows that between July 2022 to June 2023, unlicensed CBPM prescriptions surged to 177,556. In all of 2023, 282,920 prescriptions were issued for unlicensed cannabis medicines and in 2024, the number of prescriptions rose by 133% to nearly 660,000.
This underscores robust patient demand and the central role of private clinics in current access pathways.
Product Types and Consumption Patterns
There were more than 300 different medical cannabis products available in the UK and over 40 private clinics providing medical cannabis treatment, as of October 2024. According to May 2025 data from Prohibition Partners, close to 72% of medical cannabis products available on the UK market are in flower form (nearly all of them with high THC content). Extracts account for 22.5%, followed by vapes (4.9%) and edibles (0.9%).
From 2022 to 2024, the total amount of medical cannabis flower prescribed to patients surged by 262% from 2,700 kg to nearly 10,000 kg, indicating strong growth in patient numbers and possibly larger average prescription sizes. With already more than one million grams of flower prescribed in January and February of 2025, this growth is expected to continue.
Notably, import data reveals even greater underlying demand. Between January 2023 to September 2023, the UK imported 23,890 kg of cannabis (including CBPMs in flower form) which is more than double the amount prescribed to patients for all of 2024.
Clinical Evidence and Research
In 2019, the UK launched a registry study Project Twenty21, aiming to monitor the health outcomes of 20,000 patients using cannabis based medicinal products by the end of 2021. The project gives access to medical cannabis for patients living with:
- Anxiety Disorder
- Chronic Pain
- Multiple Sclerosis (MS)
- Post-Traumatic Stress Disorder (PTSD)
- Substance Use Disorder (as a harm reduction strategy)
- Tourette’s Syndrome.
The study revealed that around 49% of patients use medical cannabis for pain conditions, 42% use it for psychiatric conditions, 6% use it for neurological conditions and 3% use it for other conditions.
As of December 2022, 20 randomised clinical trials for cannabis-based medicines have been completed since 2018, and 13 were still ongoing.
Domestic Production and International Supply
While the UK is building its domestic cultivation capacity, supply remains largely import-dependent. As of November 2025, 16 companies hold licenses to cultivate high-THC cannabis in the UK.
In March 2023, Celadon Pharmaceuticals became the first British medical cannabis manufacturer to be licensed by the Home Office to sell its products in the UK after its 100,000 sq ft manufacturing facility in Birmingham was granted a Good Manufacturing Practice registration by the UK’s Medicines and Healthcare products Regulatory Agency in January.
The largest importer of cannabis into the UK in 2024 was Spain (4,254 kg), followed by Canada (2,513 kg), Portugal (2,466 kg), and Australia (2,182 kg). Germany and North Macedonia also make notable contributions to the UK’s cannabis supply.
Beyond the domestic market, there are significant opportunities present in the international industry. Life Sciences is one of the UK’s top exporting sectors, valued at £28.1 billion in 2022. which positions the country well to become a leading exporter of medical cannabis. With growing regulatory maturity, manufacturing capability, and clinical infrastructure, medical cannabis could emerge as a strategic export product.
UK Cannabis Market Infographics
How Can Cannabusinessplans Templates Help Start or Grow Your Cannabis Business?
The majority of cannabis licensing programs require the submission of a business plan as part of the application for a cannabis license. Even if it is not mandated by law, a business plan is essential for securing funding and assisting an applicant in understanding how much money it will take to start a cannabis business and how much profit it could make.
A well-structured business plan can make all the difference in helping you demonstrate your proposed cannabis business’ compliance with regulations and its operational viability.
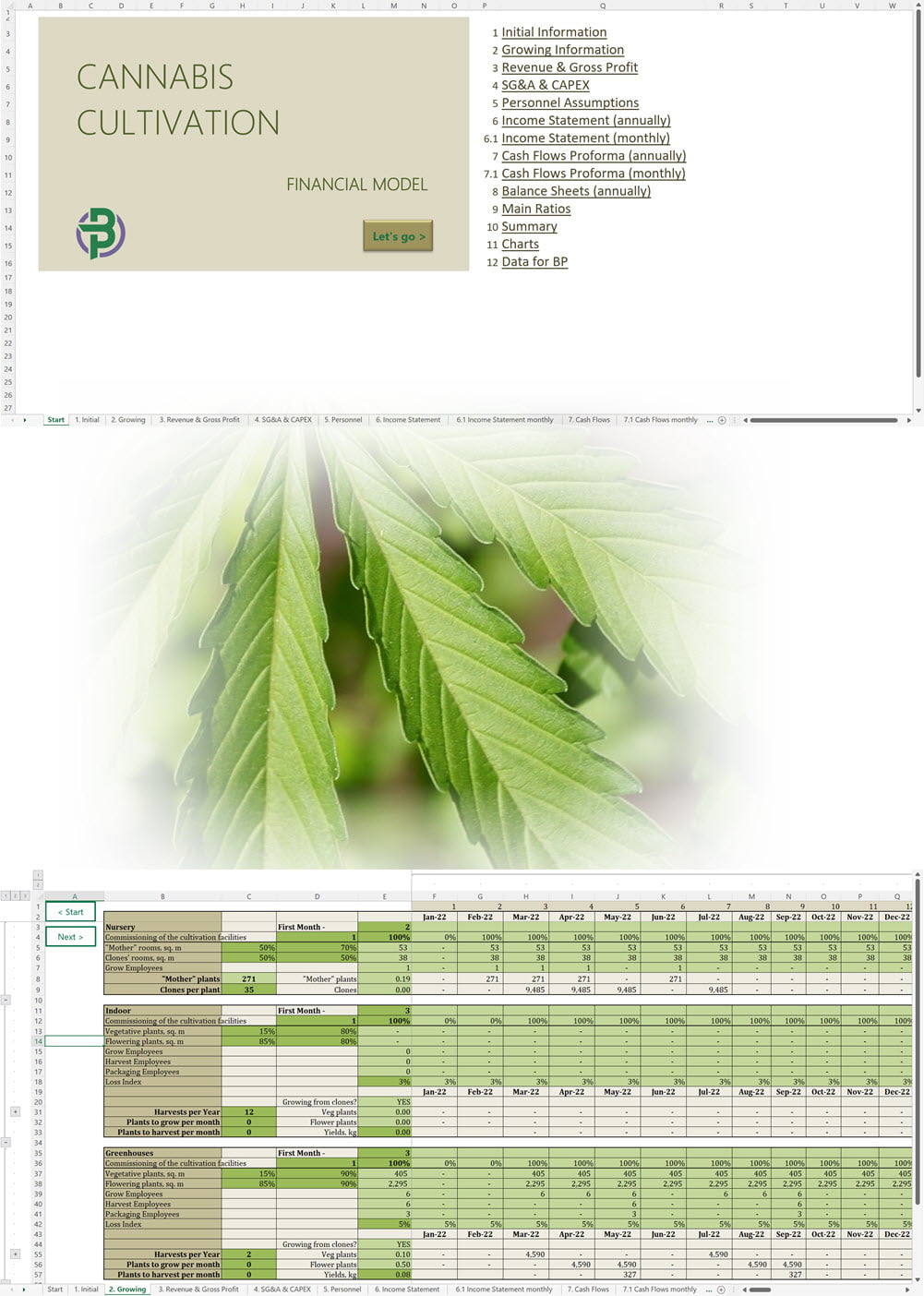
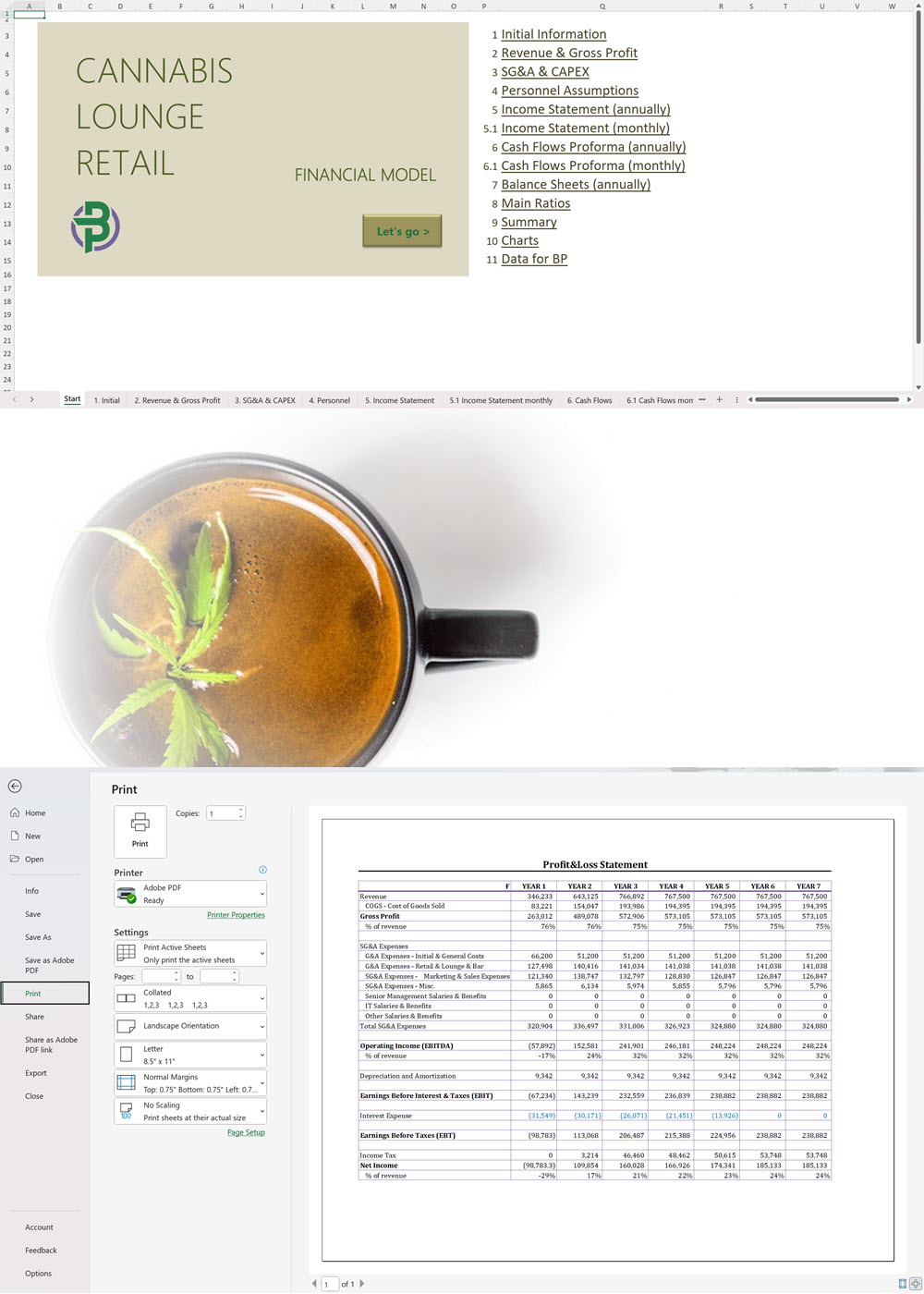
Our complete cannabis business plan template package is everything you need to create a professional business plan for a cannabis business with expert financials and projections. A complete cannabis business plan template package includes:
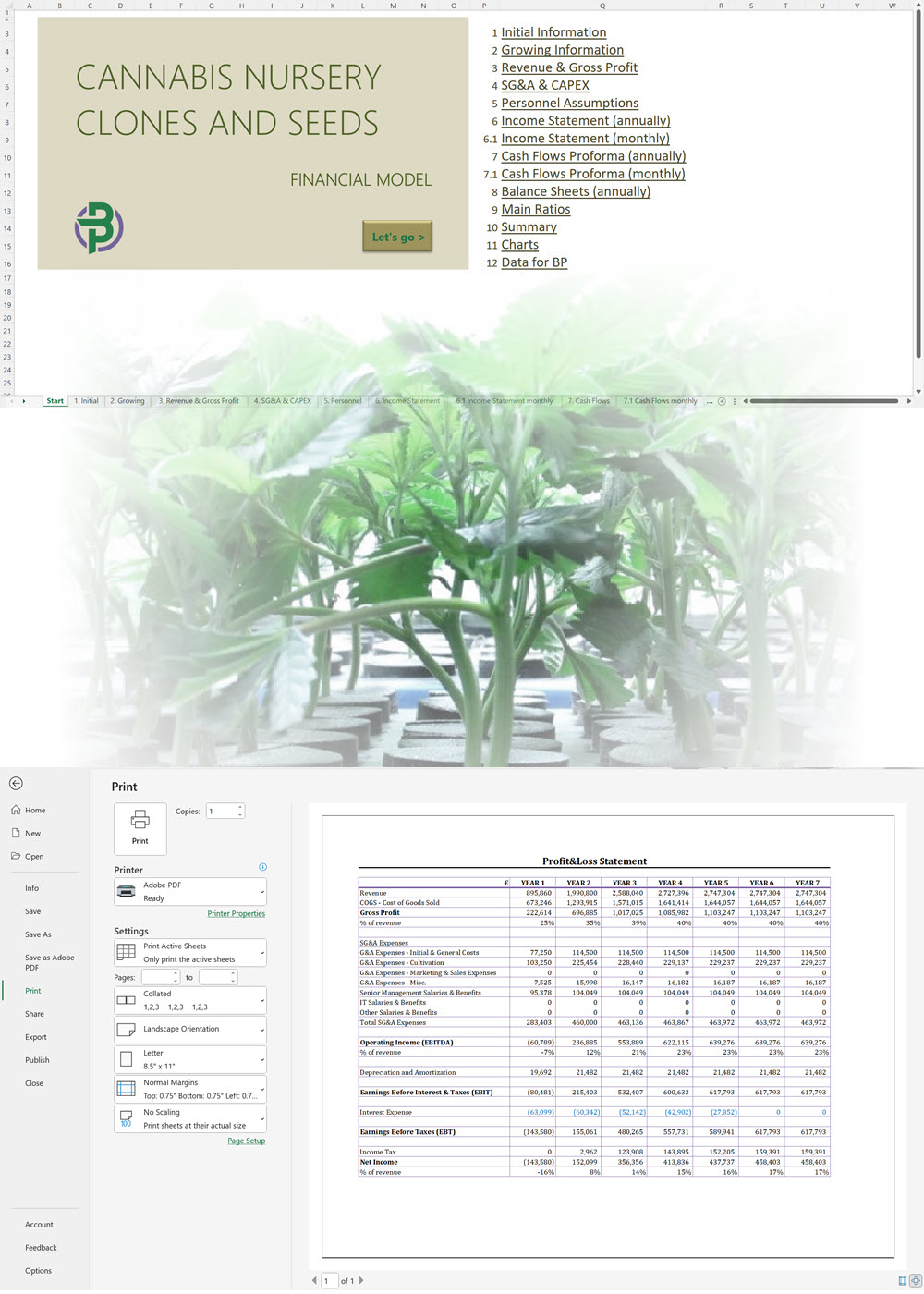
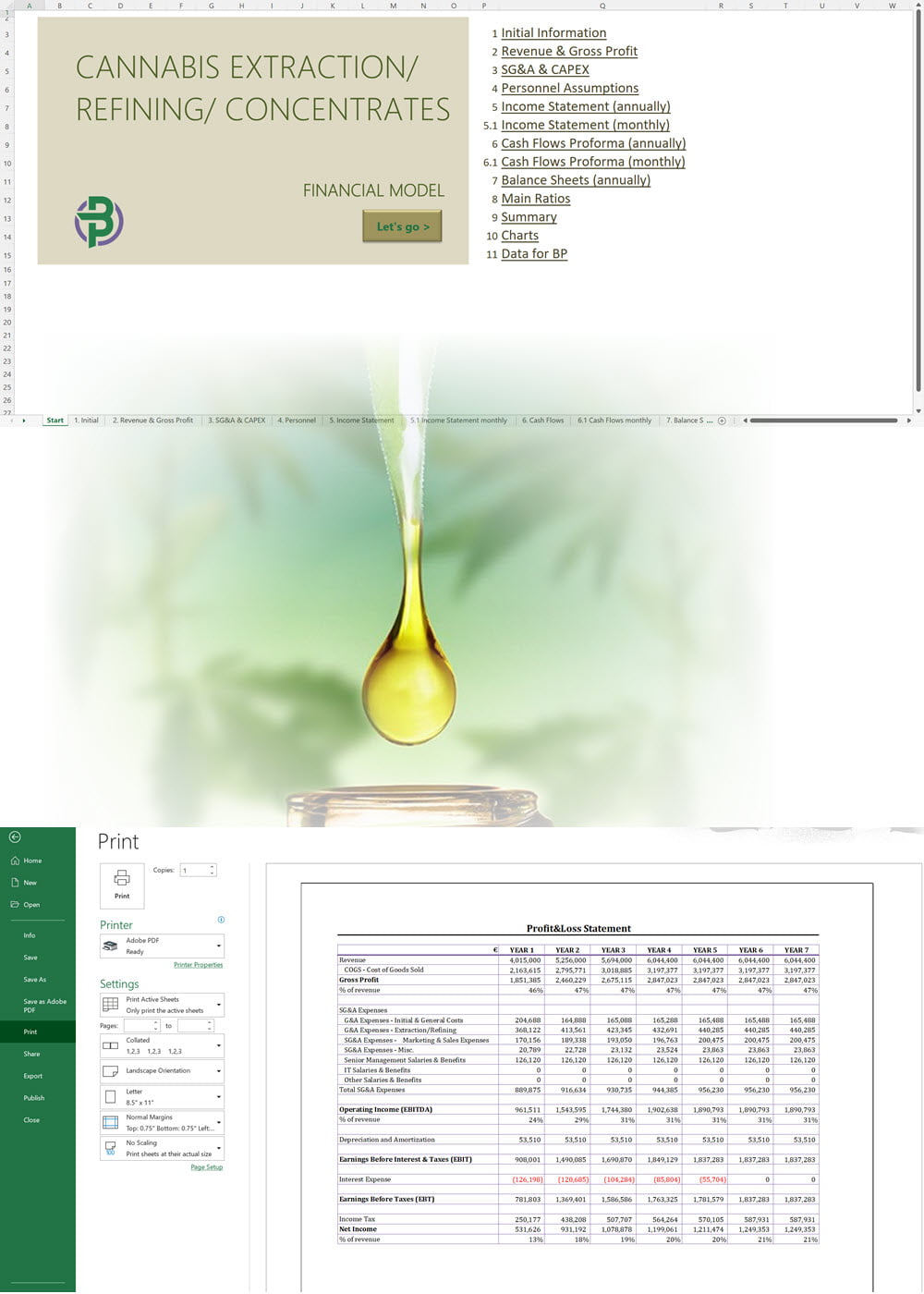
- Excel Financial Model: change variables and immediately see the impact, break down operational and capital costs, know how much it will take to get into the business and the potential profits.
- Word Business Plan: value proposition, market analysis, marketing strategy, operating plan, organizational structure, financial plan and more.
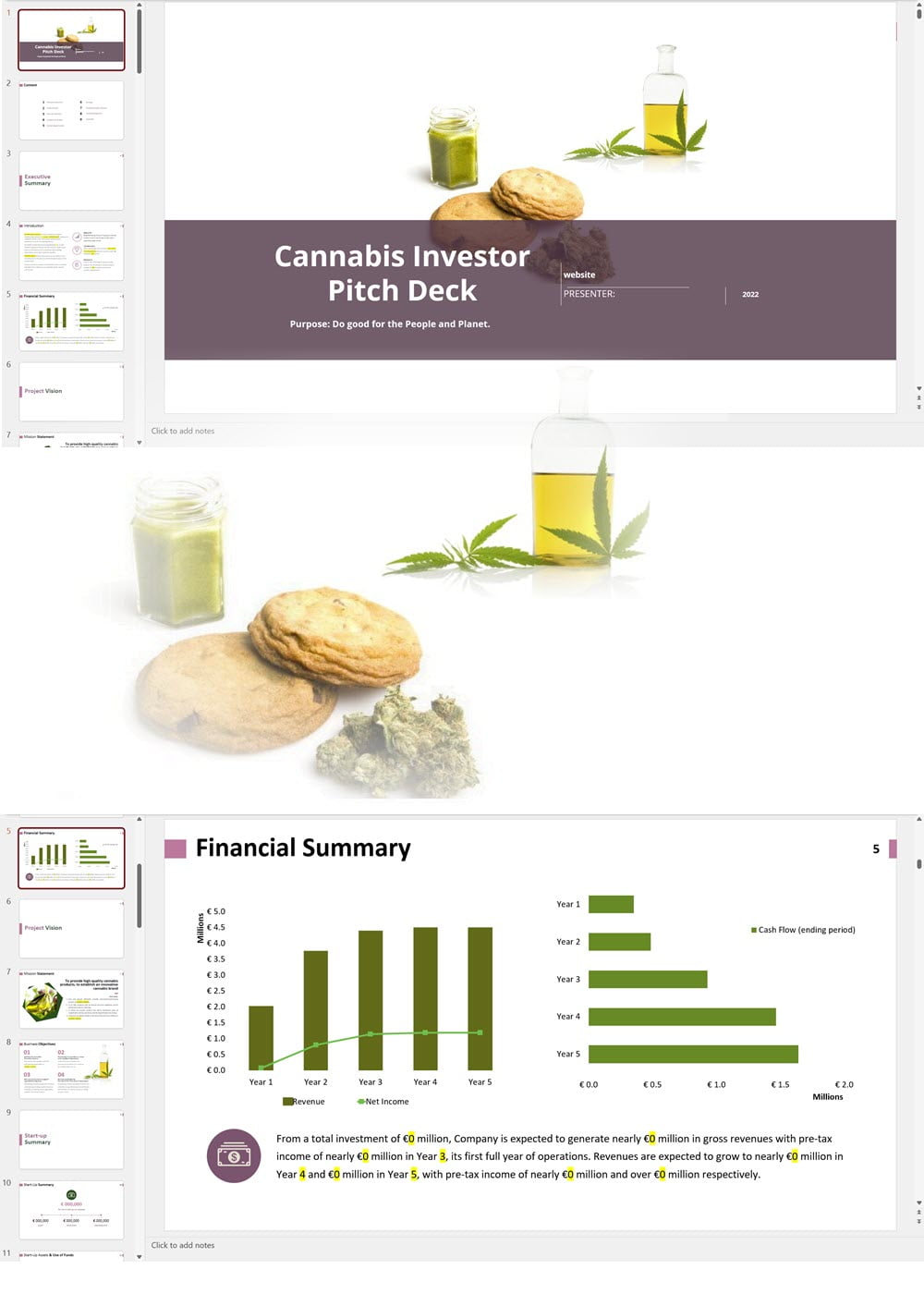
- PowerPoint Pitch Deck: provide a quick overview of your cannabis business plan.
'70% ready to go' business plan templates
Our cannabis financial models and cannabis business plan templates will help you estimate how much it costs to start and operate your own cannabis business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates.
Cannabis Retail and/or Lounge Investor Pitch Deck Sample, the UK
Best Selling Templates
-

Cannabis Cultivation Business Plan Template
Price range: €75 through €350 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Dispensary Investor Pitch Deck Template
€75 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Financial Model All in One
€250 Select options This product has multiple variants. The options may be chosen on the product page
Cannabis business plan templates for other countries are available at cannabusinessplans.com.