Medical Cannabis Legalization
Medical cannabis use was legalized in Greece in June 2017 – the sixth EU country to do so – and on 2 March 2018, the Greek government introduced Law 4523/2018 and announced plans to cultivate cannabis for domestic consumption and export.
In May 2018, the Joint Ministerial Decree No. 51483/700/Φ.15 outlined the terms and conditions for the cultivation and processing of medicinal cannabis. Companies must be vertically integrated and licenses are nontransferable.
According to the law passed in 2018, the license for the cultivation and processing of medical cannabis in Greece has two main stages:
- 1st stage: obtaining the first license which is called installation approval, which is the license that allows the medical cannabis investor to start building the facility.
- 2nd stage: as soon as the facility is designed and built, it is necessary to get the operation approval which certifies that everything is done properly and according to the EU-GMP standards.
In May 2021, the Greek Parliament voted in favor of Law 4801/2021 to legalize production, export and distribution of final products of pharmaceutical cannabis of the species Cannabis Sativa containing over 0.2% Tetrahydrocannabinol (THC). The exported medical cannabis product must have the authorization necessary to be marketed only in the country where it is being imported – no marketing authorization from Greece is required. In November 2021, the Greek government introduced a ban on the importation of cannabis products.
In March 2022, the terms and conditions for the license to export medical cannabis products in the form of dried flower were stated through the Joint Ministerial Decree No. 27462/2022.
In Greece, patients can be prescribed medical cannabis for:
- Prevention and treatment of severe nausea or vomiting caused by chemotherapy, radiotherapy and combination therapy for HIV or hepatitis C.
- Treatment of chronic pain, related to cancer or diseases of the central or peripheral nervous system, such as neuropathic pain caused by: nerve damage, “phantom limb”, trigeminal neuralgia, postherpetic neuralgia.
- Treating spasticity associated with multiple sclerosis or spinal cord injuries.
- Use as an orexigenic in palliative care of patients undergoing treatments for cancer or acquired immunodeficiency (AIDS).
Medical cannabis can be prescribed by anesthesiologists, neurologists, pathologists – oncologists, pathologists – infectious disease specialists, and rheumatologists.
To be able to use medical cannabis, patients must first obtain a prescription through the electronic platform from their doctor. The patients can then pick up the medical cannabis preparation prescribed by their doctor from a pharmacy.
A special marketing authorization must be obtained from the National Medicines Agency (EOF) in order to be able to make a medical cannabis product available for patients in Greece.
How to Start a Cannabis Business in Greece?
Those wishing to cultivate, transport, process, supply or export medical cannabis and final medicinal cannabis products with a THC content of more than 0.2% must obtain a license.
Installation approval application fee: €10,000
Operation approval application fee: €10,000
If the applicant proceeds with a single approval for installation and operation, then a fee of €15,000 is required.
Licensing Process:
- A completed questionnaire found in Annex I of the Joint Ministerial Decree No. 27462/2022 must be submitted to the Business Licensing and Business Parks Department of the General Secretariat for Industry (GGB Licensing Department).
- The GGB Licensing Department shall provide the applicant with a response within 5 working days, stating the required supporting documents for the installation approval and the operation approval, respectively.
- The applicant submits the supporting documents for the installation approval in six copies: one in printed form and the remaining five in digital form.
- The Licensing Directorate of the General Secretariat of the Hellenic Republic transmits to the competent Services of the Ministry of Rural Development and Food, the Ministry of Health/EOF and the Hellenic Police, a digital copy accompanied by a copy of the submitted questionnaire. Within 30 days, the EOF will issue an opinion regarding the granting of the establishment approval.
- Within 30 days from the submission of a complete file, the installation approval is granted by a joint decision of the competent authorities of the bodies of the Ministries of Development and Investments, Health and Rural Development and Food. This approval is valid for 5 years.
- Within 15 days of granting the installation approval, the Licensing Department of the General Secretariat of the Hellenic Republic shall conduct an on-site inspection to confirm the suitability of the space and the correctness of the questionnaire.
- The applicant submits the supporting documents for the operation approval must be submitted in six copies: one in printed form and the remaining five in digital form.
- The Licensing Department of the General Secretariat of the Hellenic Republic of the Hellenic Republic transmits a digital copy to the competent Services of the Ministry of Rural Development and Food, the Ministry of Health/EOF and the Hellenic Police. Within 30 days, the EOF will issue an opinion regarding the granting of the establishment approval.
- Within 30 days from the submission of a complete file, the operation approval is granted by the decision of the Ministers of Economy and Development, Health and Rural Development and Food. The approval is valid for 10 years.
- Within 2 months of the issuance of the operation approval, the Licensing Department of the General Secretariat of the Hellenic Republic shall conduct an on-site inspection to confirm that the conditions for granting the operation approval have been satisfied.
Supporting documentation to be submitted:
- Land use certificate from the competent Building Service along with a topographic map used for its issuance.
- Environmental Impact Study and Environmental Terms Approval Decision.
- Technical report of the entity documenting the the capacity of the hemp processing unit is enough to the utilization of the produced quantity of hemp cultivated in the facility. The report must also contain a floor plan of the area occupied by the processing unit.
- Copy of identity card or valid passport of the applicant or, if the applicant is a legal entity, those involved in the administration and management of the business.
- Copy of the general criminal record of the applicant or, if the applicant is a legal entity, those involved in the administration and management of the business.
- Solemn declaration regarding compliance with requirements a, b and c of article 2Z of Law 4139/2013.
- Certificate of non-bankruptcy, non-filing of a bankruptcy application, non-filing of an application for conciliation-reorganization, non-filing of an application for dissolution and non-dissolution of the legal person.
- Tax compliance certificate and insurance certificate of the applicant issued no later than one month before the date of submission of the application.
- Solemn declaration that there is no school within a distance of less than 1,000m from the boundaries of the proposed cannabis establishment.
- A certified title deed or a lease agreement or a free concession endorsed by the competent Tax Office.
- A diagram of the coverage of an enclosed area showing all building, mechanical and other installations, as well as a floor plan of greenhouse areas with a depiction of the cultivated area and the processing areas with an indicative placement of the machinery within them.
- A certificate from the relevant Security Sub-Directorate or the Security Department that the terms and conditions of safe storage have been complied with regarding the facilities.
- Solemn declaration of a legally competent engineer regarding the static adequacy, the industrial use of the building, the installation of mechanical equipment in a main use area, the number of the existing building permit as well as the non-requirement to issue a new one.
- Solemn declaration stating how many and which specialty technicians it will use by law.
- Solemn declaration of engineers of the respective specialty, that the installation was carried out in accordance with the granted installation approval.
- Solemn declaration of a legally competent engineer that the fire protection measures have been taken for the installation of the processing unit and that they are functioning properly.
- Solemn declaration regarding compliance with the requirements of article 2Z of Law 4139/2013 for those associated with any employment relationship with the unit.
- Approval of the cutting-down of the sidewalk level or approval of a traffic connection or entry-exit approval from the competent Service for the maintenance of the road (or roads) in front of the unit, accompanied by a solemn declaration from a legally competent engineer that the traffic connection or the entry-exit of the facility was carried out in accordance with the approved plans.
Some additional documentation may be required – these can be found on the official website of the National Registry of Administrative Procedures.
The Licensing Department of the General Secretariat of the Hellenic Republic shall conduct on-site inspections at least annually to check that the conditions for the installation and operation approval are being satisfied.
By 31 December of each year, the licensee must submit updated supporting documentation for the installation and operation approval.
A special permit is required for the installation of greenhouses and greenhouse-type chambers for medicinal cannabis. The process to obtain it is outlined here.
No extra license is required for the import or export of propagating material of cannabis varieties with a THC content of more than 0.2%.
There are also production licenses available exclusively for the export of final medicinal cannabis products or finished medicinal cannabis products in the form of dried flower specifically.
To obtain this license, an application must be submitted to the EOF, accompanied by the following:
- An official translation of the license or other document of the competent authorities of the country to which the final medicinal cannabis product will be exported, evidencing:
- That the applicant is permitted to import the products into that country.
- Description of the produced medicinal cannabis product.
- Its qualitative and quantitative composition, as well as THC content.
- Its purpose of use.
- Its form and packaging.
- A description of the production process and validation of its reliability.
- A description of the method of analysis of the final produced medicinal cannabis product and validation of its reliability.
Fee: €2,500 (application) and €400 per form and strength.
Within 90 days from the submission of a complete file, the EOF will grant the license.
To obtain this license, the applicant must submit to the EOF the installation and operation approvals as well as the following:
- Documented agreement of the cannabis flower with the general texts of the European Pharmacopoeia and reference to methods, microbiological quality, aflatoxins, heavy metals, pesticides, pyrrolizidine alkaloids or, in the absence thereof, with the general texts of a national pharmacopoeia of another EU member state or a pharmacopoeia of a third country, such as the USA.
- Description of the production process and validation of its reliability.
- Certificate of the Directorate for Licensing of Enterprises and Business Parks of the General Secretariat for Industry.
The applicant must also meet the safety and production capacity requirements of the processing units producing medicinal cannabis products stated in Article 15 of the Ministerial Law No. 4801/2021.
In order to be able to sell a final medicinal cannabis product through pharmacies in Greece, a marketing authorization is required from the EOF. This is necessary both for a product produced in Greece and for a product imported into Greece. The marketing authorization is valid for 3 years.
An application for a marketing authorization shall be accompanied by the following information and documents:
- Name or company name and residential address or registered office of the applicant.
- Name of the final medicinal cannabis product.
- Qualitative and quantitative composition of the final medicinal cannabis product.
- Methods and specifications for raw material quality control and validation of the control methods.
- Development studies of the final medicinal cannabis product.
- Production process, necessary intermediate quality controls during production, critical stages of the production process and validation of the production process in compliance with guidelines of good manufacturing for medicinal products intended for human use as defined by Greek legislation.
- Methods and specifications for quality control of the final product, validation of the control methods, results of analysis of test batches.
- Composition, type, characteristics of the immediate container.
- Stability studies of the final product.
- Therapeutic indications, contraindications and adverse reactions.
- Dosage, pharmaceutical form, method and route of administration and expected shelf life of the product.
- Justification for any precautionary and safety measures to be taken for the storage of the product, its administration to patients and the disposal of residues, as well as the potential risks that the medicinal product presents for the environment.
- A summary of the applicant’s adverse reaction monitoring system which must include:
- Proof that the applicant has a qualified person responsible for the monitoring of adverse reactions.
- Contact details of the qualified person.
- Risk management plan.
- Summary of the product characteristics including:
- Maximum quantitative composition limit in THC.
- Pharmaceutical form.
- Pharmaceutical properties.
- A mock-up of the outer packaging and primary packaging of the medicinal product.
At least 9 months prior to the expiration date of the marketing authorization, the holder of the marketing authorization must submit all the data of the adverse reaction monitoring system file.
Cannabis Market in Greece
With its favorable climate and geographical location, Greece can play a leading role in the development of the medical cannabis industry. According to a 2020 study, a medical cannabis operation that owns a 1 hectare greenhouse and cultivates and processes medical cannabis in Greece would earn net profit of €3.5 million in the first year of operations and around €7.07 million annually from the second to the tenth year.
From 140 approved facilities to date, only 4 have received operating approval and only one of them has received authorization to place its products on the market.
In February 2024, the first locally produced finished medicinal cannabis products – preparations of dried cannabis flower – became available to patients with a prescription. It is estimated that one of the new medicines has been administered to around 4,000 patients in 2024 and another 4,000 patients already in the first quarter of 2025.
In December 2024, Tikun Europe made the first export of finished medical cannabis products from Greece to Switzerland. Tikun continued to expand its export activity in 2025, exporting medicinal cannabis to the UK and German markets in May 2025.
According to the European Cannabis Report, by 2028 Greece’s industrial and pharmaceutical cannabis industry could inject some €2 billion into the local economy.
How Can Cannabusinessplans Templates Help Start or Grow Your Cannabis Business?
The majority of cannabis licensing programs require the submission of a business plan as part of the application for a cannabis license. Even if it is not mandated by law, a business plan is essential for securing funding and assisting an applicant in understanding how much money it will take to start a cannabis business and how much profit it could make.
A well-structured business plan can make all the difference in helping you demonstrate your proposed cannabis business’ compliance with regulations and its operational viability.
Our cannabis business plan template package is everything you need to create a professional business plan for a cannabis business with expert financials and projections. A complete cannabis business plan template package includes:
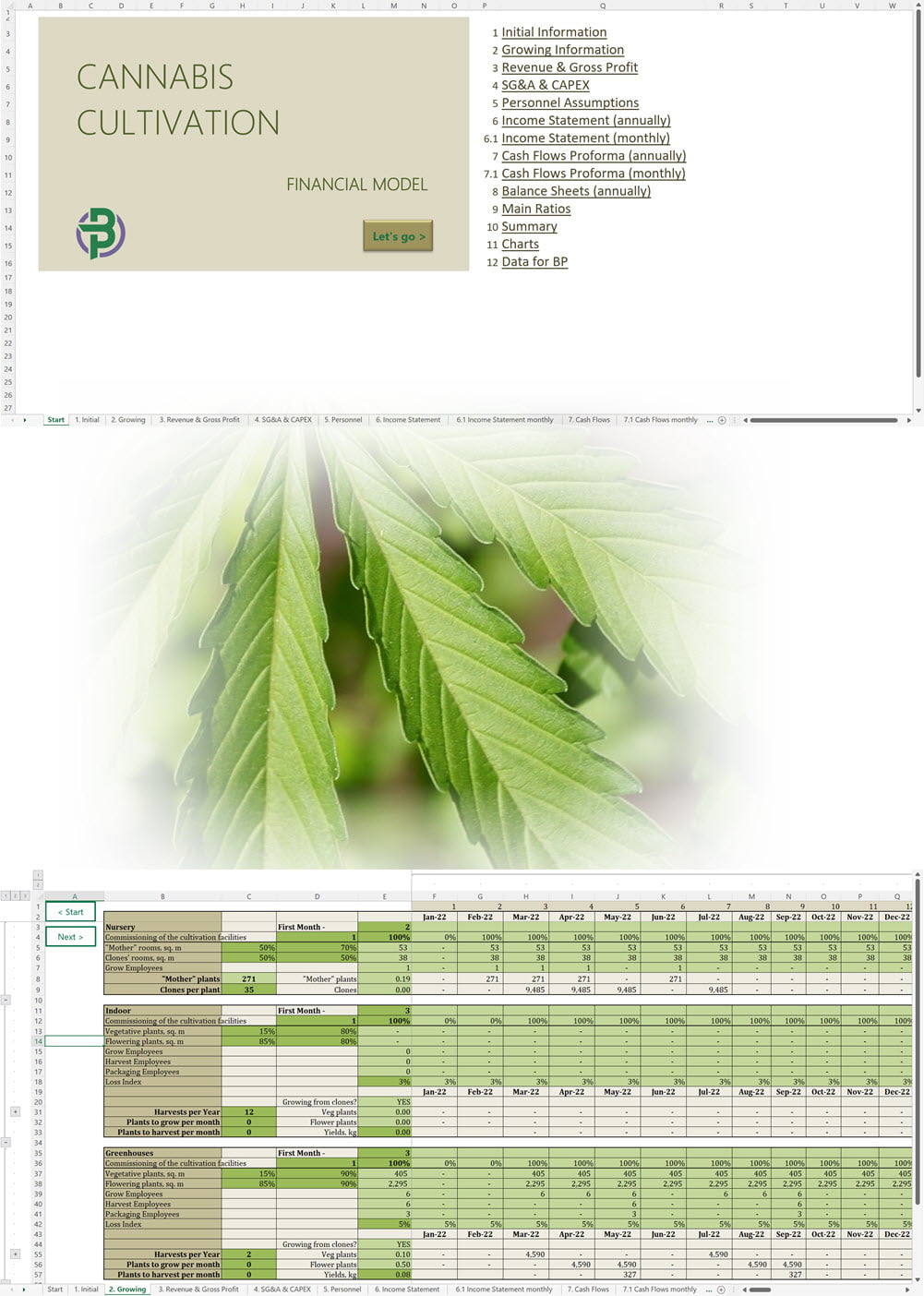
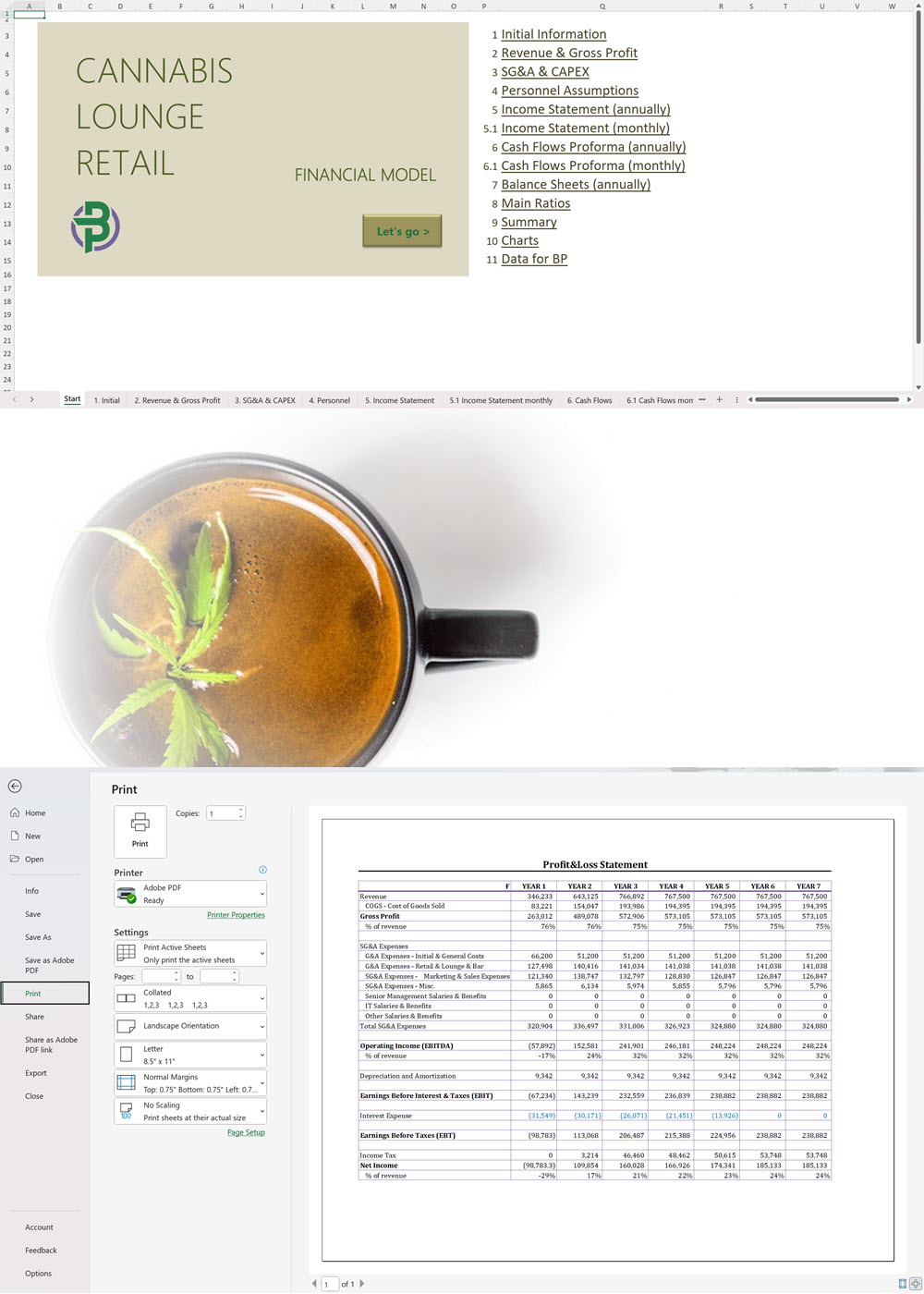
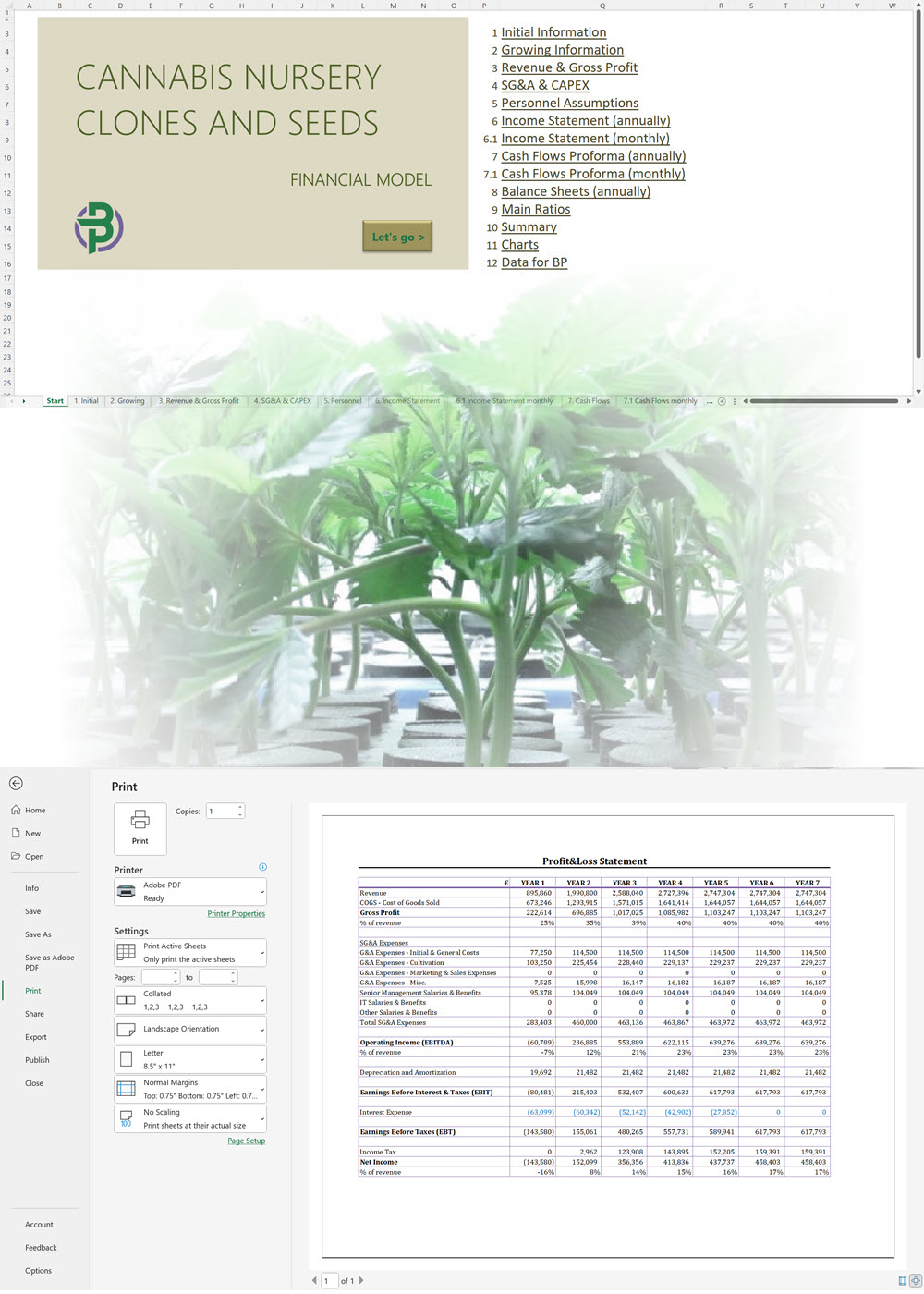
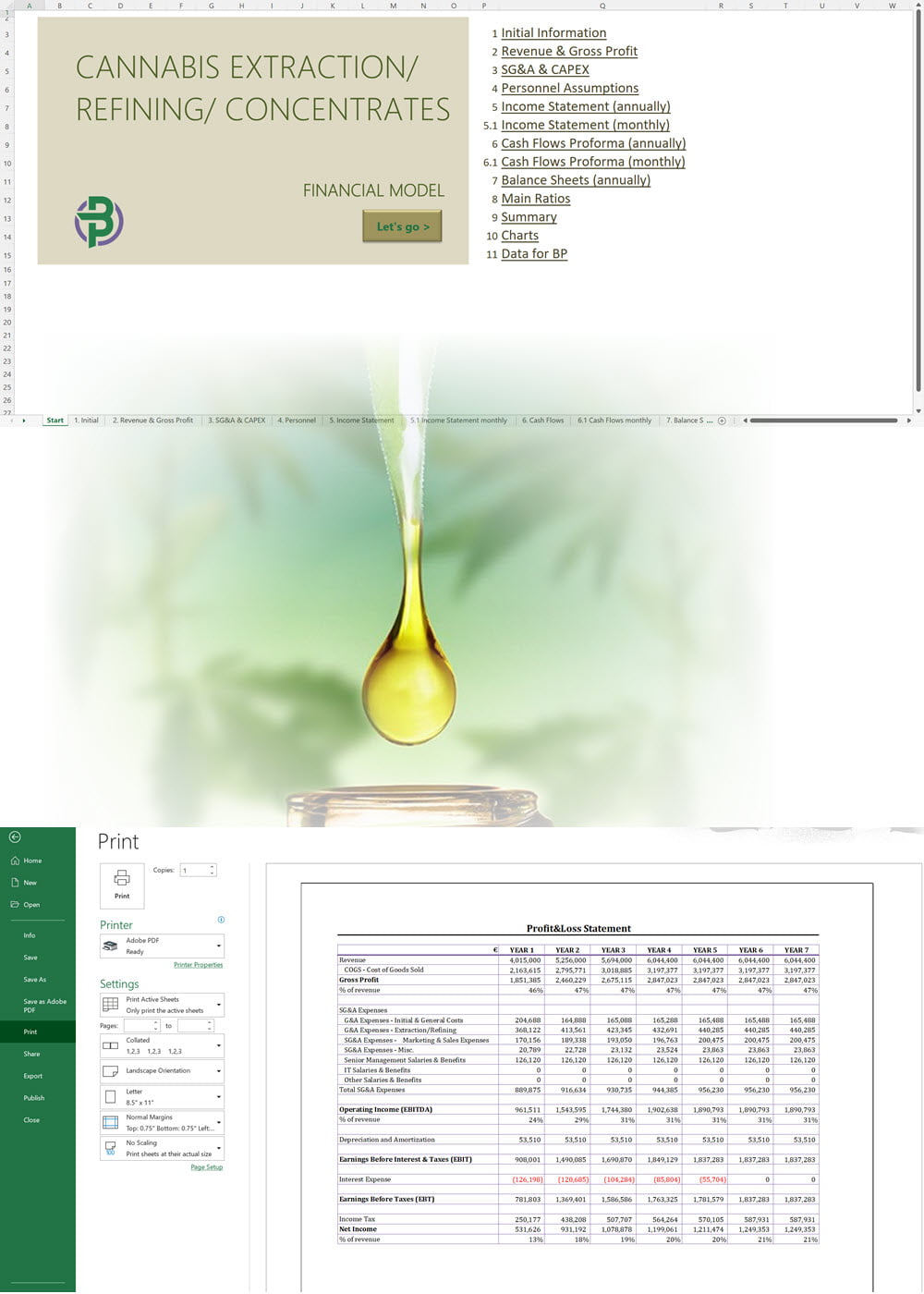
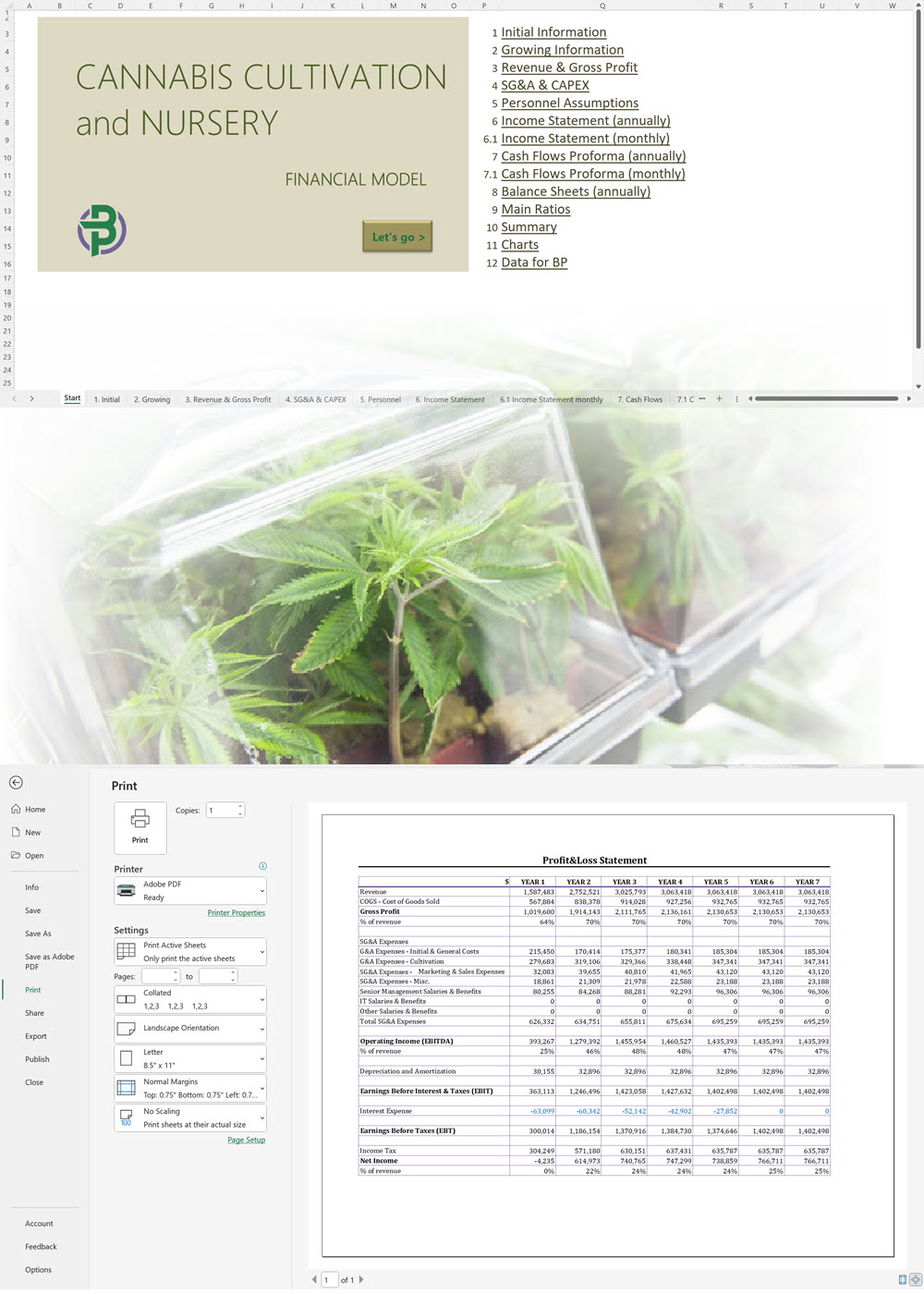
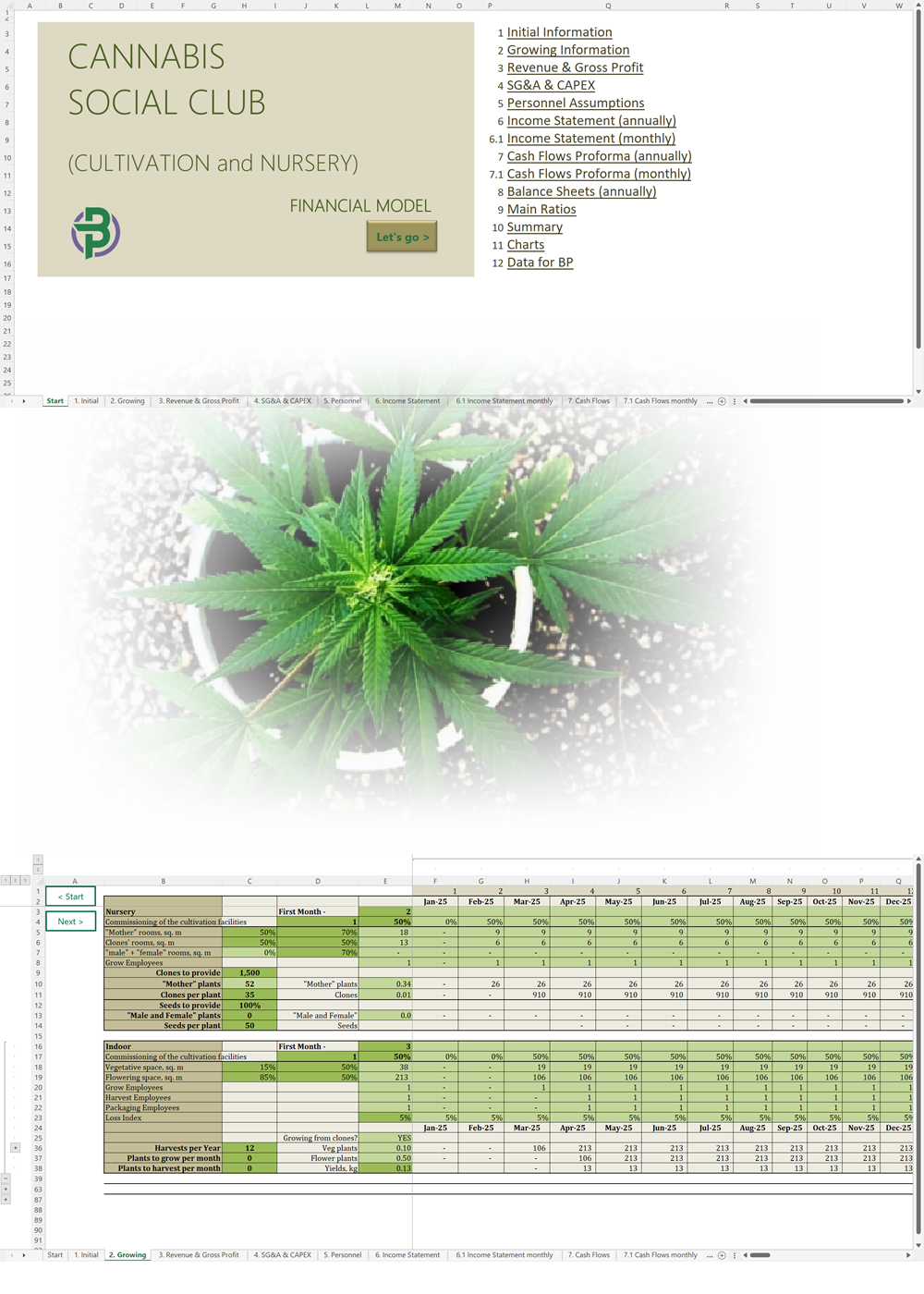
- Excel Financial Model: change variables and immediately see the impact, break down operational and capital costs, know how much it will take to get into the business and the potential profits.
- Word Business Plan: value proposition, market analysis, marketing strategy, operating plan, organizational structure, financial plan and more.
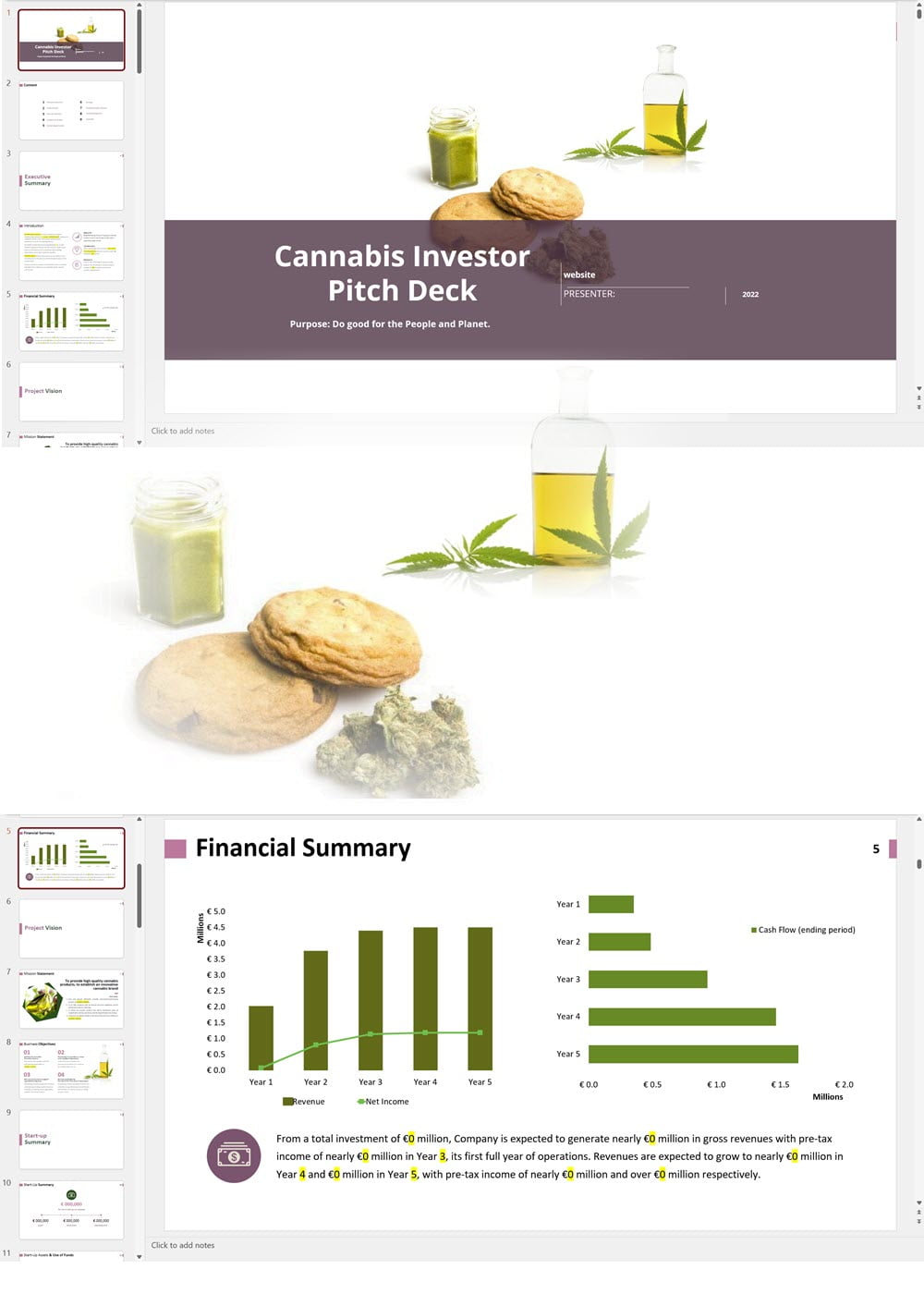
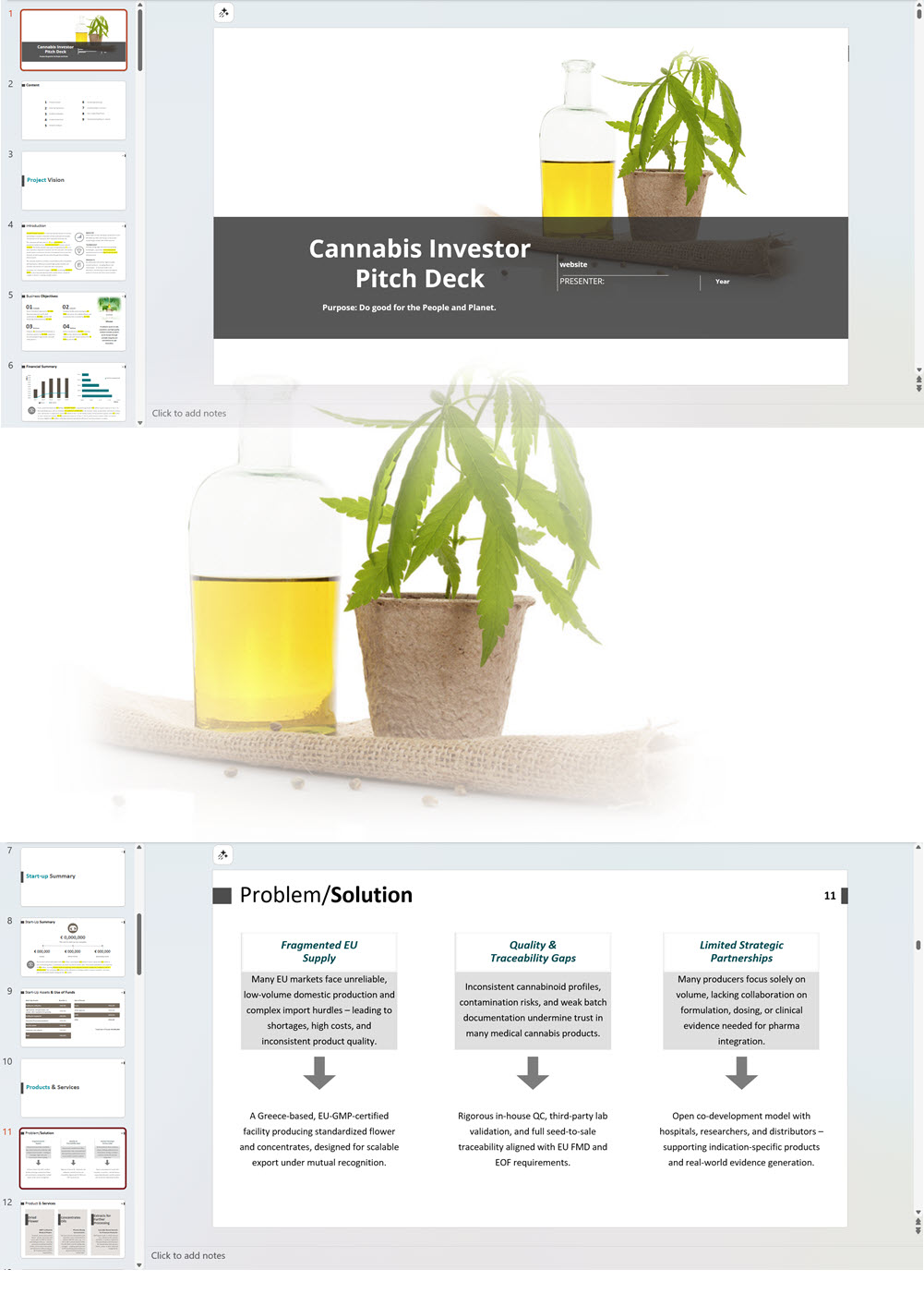
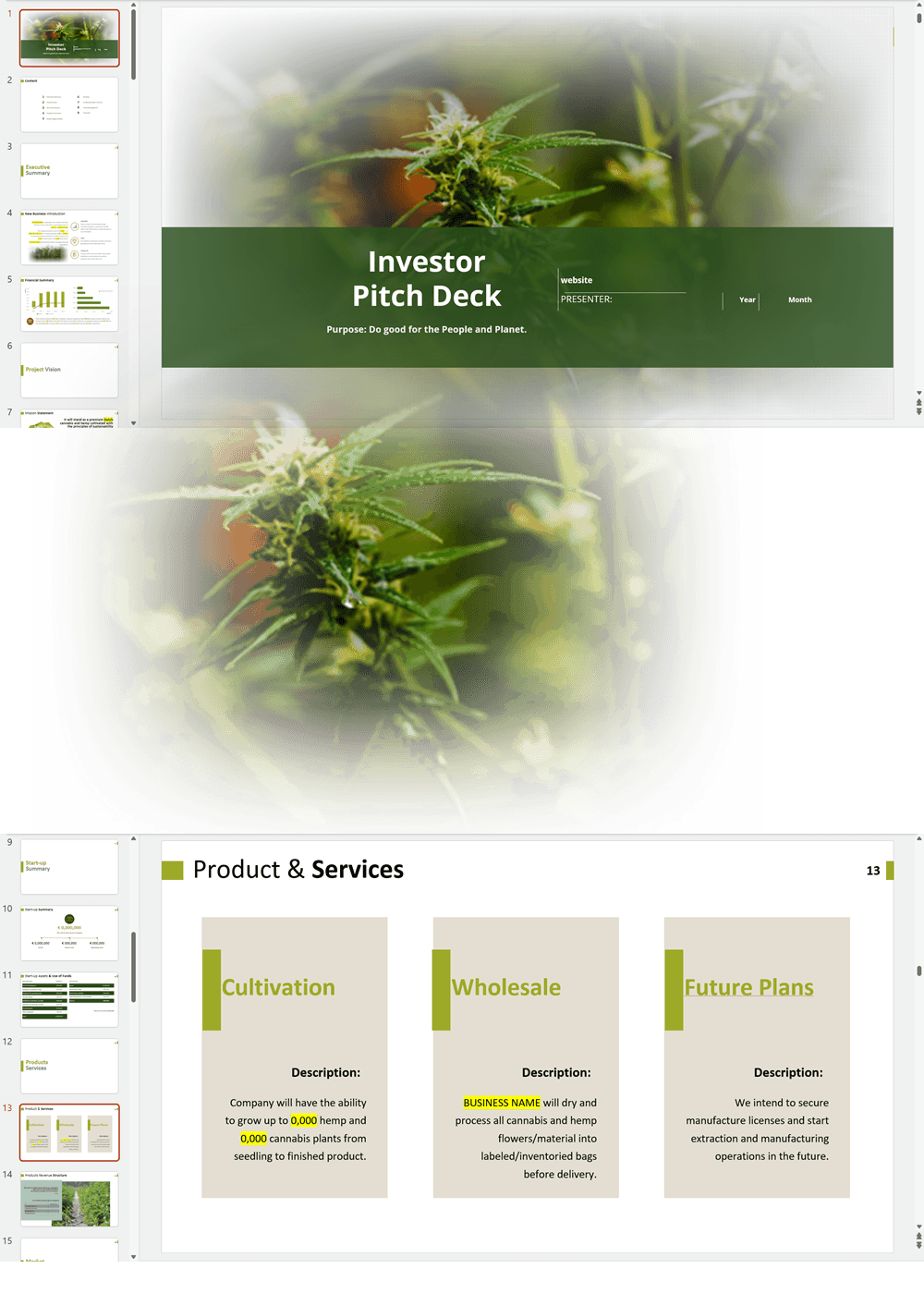
- PowerPoint Pitch Deck: provide a quick overview of your cannabis business plan.
- Unlimited support!
'70% ready to go' business plan templates
Our cannabis financial models and cannabis business plan templates will help you estimate how much it costs to start and operate your own cannabis business, to build all revenue and cost line-items monthly over a flexible seven year period, and then summarize the monthly results into quarters and years for an easy view into the various time periods. We also offer investor pitch deck templates.
Cannabis Cultivation and Extraction Business Plan Sample, Greece
Best Selling Templates
-

Cannabis Cultivation Business Plan Template
Price range: €75 through €350 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Dispensary Investor Pitch Deck Template
€75 Select options This product has multiple variants. The options may be chosen on the product page -

Cannabis Financial Model All in One
€250 Select options This product has multiple variants. The options may be chosen on the product page
Templates for other countries are available at cannabusinessplans.com.